Applied Chemistry I: UNIT 2: Nanochemistry
Preparation of Nanomaterials
The following two approaches are used for the synthesis of nanomaterials. 1. Top‒down process (or) Physical (or) Hard methods. 2. Bottom‒up process (or) Chemical (or) Soft methods.
PREPARATION OF NANOMATERIALS
The following two
approaches are used for the synthesis of nanomaterials.
1. Top‒down process
(or) Physical (or) Hard methods.
2. Bottom‒up process
(or) Chemical (or) Soft methods.
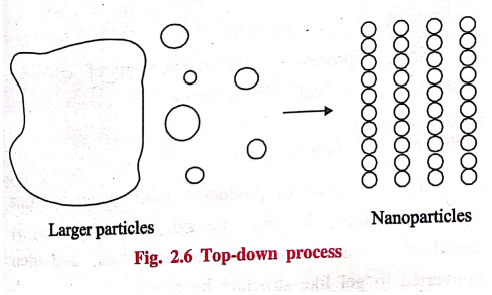
1. Top‒down process
Top‒down process
involves the conversion of bulk materials into smaller particles of nano‒scale
structure.
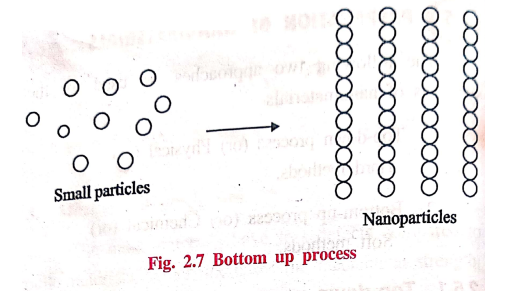
2. Bottom‒up process
Bottom‒up process
involves building‒up of materials from the bottom by atom by atoms, molecule by
molecule (or) cluster to the nanomaterials.
3. Important Preparations
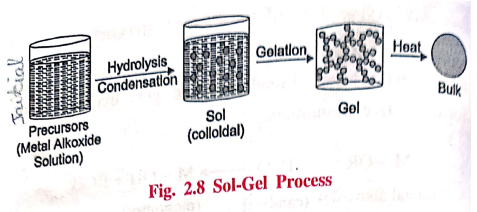
1. Sol‒Gel process
Definition
Sol‒gel process is the
conversion of colloidal solution (sol) to "gel" like structure.
Principles of Sol‒gel process
It is a method of
producing solid materials from small molecules. In this method, the precursor
is dissolved in water (or) alcohol to form "sol" and then converted
to gel like structure by heating.
Sol‒gel process is a
wet chemical technique. Generally, it is used to produce metal oxides.
Preparation: Preparation of metal oxide sol‒gel
Preparation of sol‒gel
involves the following steps.
Step
1: Preparation of monomers (precursor)
Metal alkoxide is
dissolved in alcohol and then diluted with water. Metal alkoxide gets
hydrolysed to form reactive monomers.
M‒OR (metal alkoxide) +
H2O (catalyst) → M‒OH (monomer) + ROH
Step
II: Formation of "Sol"
Condensation of these
monomers to form colloid like solution (sol).
M‒OH + ROM → M‒O‒M +
ROH
M‒OH + HO‒M → M‒O‒M + H2O
Step
III: Formation of "Gel"
"Sol" gets
converted to "gel" via polycondensation.
Step
IV: Aging process
It is the process,
where condensation occurs with the gel network that can cause expulsion of
solvent.
Step
V: Drying
It removes liquid
phases and M‒OH groups
Step
VI: Tempering
It is the process of
densification of the gel to remove the pores of the gel network.
2. Solvothermal Synthesis
Solvothermal synthesis involves
the use of solvent under high temperature (between 100°C to 1000°C) and
moderate to high pressure (1 atm to 10,000 atm) that facilitate the interaction
of precursors during synthesis.
Method
A solvent like ethanol,
methanol, 2‒propanol is mixed with certain metal precursors and the solution
mixture is placed in an autoclave kept at relatively high temperature and
pressure in an oven to carry out the crystal growth. The pressure generated in
the vessel, due to the solvent vapour, elevates the boiling point of the
solvent.
Example:
Solvothermal
synthesis of zinc oxide
Solvothermal synthesis of zinc oxide
Zinc acetate dihydrate
is dissolved in 2‒propanol at 50°C. Subsequently, the solution is cooled to 0°C
and NaOH is added to precipitate ZnO. The solution is then heated to 65°C to
allow ZnO growth for some period of time. Then a capping agent (1‒dodecanethiol)
is injected into the suspension to arrest the growth.
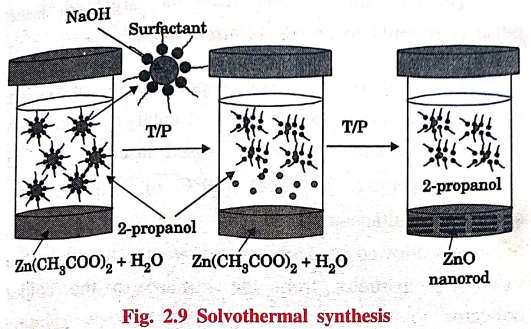
The rod shaped ZnO nano‒crystal
is obtained.
3. Laser ablation
In laser ablation
technique, high‒power laser pulse is used to evaporate the material from the
target. The stoichiometry of the material is protected in the interaction.
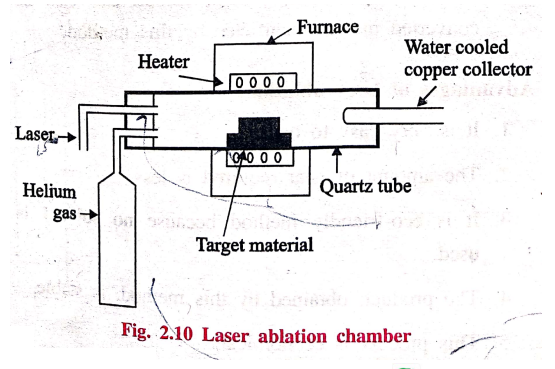
The total mass ablated
from the target per laser pulse is referred to as the ablation rate.
This method involves
vapourisation of target material containing small amount of catalyst (nickel
(or) cobalt) by passing an intense pulsed laser beam at a higher temperature to
about 120°C in a quartz tube reactor. Simultaneously, an inert gas such as
argon, helium is allowed to pass into the reactor to sweep the evaporated
particles from the furnace to the colder collector.
Uses
1. Nanotubes having a
diameter of 10 to 20 nm and 100 μm can be produced by this method.
2. Ceramic particles
and coating can be produced.
3. Other materials like
silicon, carbon can also be converted into nanoparticles by this method.
Advantages of laser ablation.
1. It is very easy to
operate.
2. The amount of heat
required is less.
3. It is eco‒friendly
method because no solvent is used.
4. The product,
obtained by this method, is stable.
5. This process is
economical.
4. Chemical Vapour Deposition (CVD)
This process involves
conversion of gaseous molecules into solid nanomaterials in the form of tubes,
wires (or) thin films. First the solid materials are converted into gaseous molecules
and then deposited as nanomaterials.
Example:
CNT
preparation.
The CVD reactor
consists of a higher temperature vacuum furnace maintained at inert atmosphere.
The solid substrate containing catalyst like nickel, cobalt, iron supported on
a substrate material like, silica, quarts is kept inside the furnace. The
hydrocarbons such as ethylene, acetylene and nitrogen cylinders are connected
to the furnace. Carbon atoms, produced by the decomposition at 1000°C, condense
on the cooler surface of the catalyst.
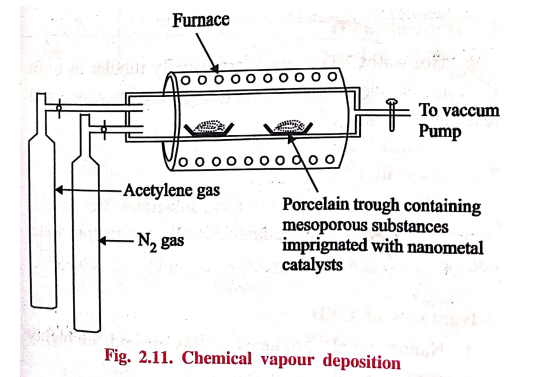
As this process is
continuous, CNT is produced continuously.
Types of CVD Reactor
Generally the CVD
reactors are of two types
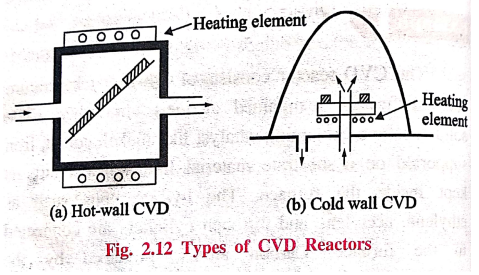
1.
Hot‒wall CVD
Hot wall CVD reactors
are usually tubular in form. Heating is done by surrounding the reactor with
resistance elements.
2.
Cold‒wall CVD
In cold‒wall CVD
reactors, substrates are directly heated inductively while chamber walls are
air (or) water cooled.
Advantages
of CVD
1. Nanomaterials, produced
by this method, are highly pure.
2. It is economical.
3. Nanomaterials,
produced by this method, are defect free.
4. As it is simple
experiment, mass production in industry can be done without major difficulties.
5. Electro‒deposition (or) Electrochemical deposition
Electro‒deposition is
an electrochemical method in which ions from the solution are deposited at the
surface of cathode.
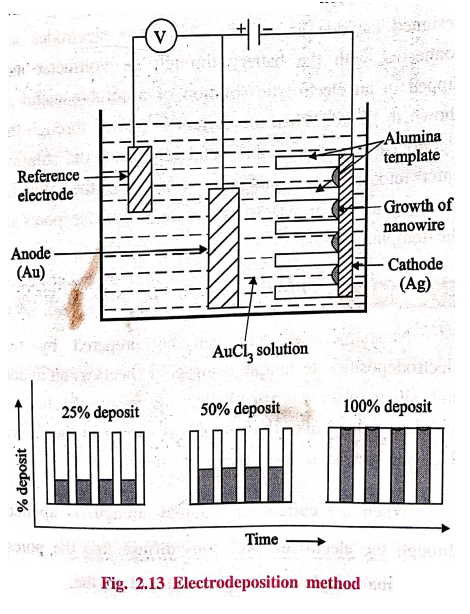
Template assisted
electro‒deposition is an important technique for synthesizing metallic nanomaterials
with controlled shape and size. Array of nano‒structured materials with
specific arrangements can be prepared by this method using an active template
as a cathode.
Process of electro‒deposition
The cell consists of a
reference electrode, specially designed cathode and anode. All these electrodes
are connected with the battery through an voltmeter and dipped in an
electrolytic solution of a soluble metal as shown in figure. When the current
is passed through the electrodes of template, the metal ions from the solution
enter into the pores and gets reduced at the cathode, resulting in the growth
of nanowire inside the pores of the template:
Example:
Electrodeposition
of Gold on Silver
Nanostructured gold can
be prepared by the electrodeposition technique using gold sheets as an anode
and silver plate as a cathode. An array of alumina template is kept over the
cathode as shown in the figure 2.13 and AuCl3 is used as an
electrolyte.
When the current of
required strength is applied through the electrodes, Au+ ions
diffuse into the pores of alumina templates and gets reduced at the cathode resulting
in the growth of nanowires (or) nanorods inside the pores of the alumina
templates.
Advantages of Electro‒deposition
1. This method is
relatively cheap and fast.
2. Complex shaped
objects can be coated.
3. The film (or) wire
obtained is uniform.
4. Metal nanowires
including Ni, Co, Cu and Au can be fabricated by this method.
6. Electrospinning
Definition
Electrospinning is a
method of producing ultrafine (in nanometers) fibres by charging and ejecting a
polymer solution through a spinneret under high‒voltage electric field and to
solidify (or) coagulate it to form a filament.
Components
1. A high voltage power
supply.
2. A polymer reservoir
that can maintain a constant flow rate of solution.
3. A conductive needle,
as polymer source, connected to the high voltage power supply.
4. A conductive
collector (plate, drum, etc.)
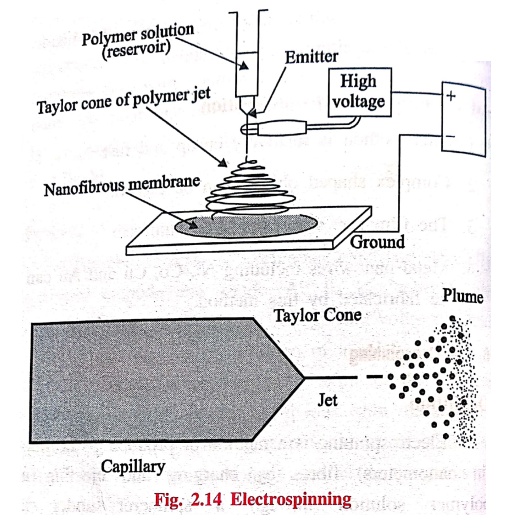
Process
A polymer is dissolved
in a suitable solvent and is filled in the capillary reservoir. When
sufficiently high voltage is applied to create an electric field between the
needle tip and the collector, a charge accumulates at the liquid surface. When
the electrostatic repulsion is higher than the surface tension the liquid
meniscus is deformed into conically shaped structure known as a Taylor cone.
Once the Taylor cone is
formed, the charged liquid jet is ejected towards the collector. Depending upon
the viscosity of the solution, solid fibre will be formed as the solvent
evaporates.
Applications
1. Electrospinning is
used in diagnosis and treatment of diabetes.
2. Electrospun fibres
are used in energy storage devices such as, solar cell, fuel cell, super
capacitors.
3. It is also used in
textiles for smart clothing, protecting clothing and fire retardant fibres.
4. It is used in
sensors like gas sensors, chemical sensors and fluorescence sensors.
5. In biomedical, it is
used in drug delivery, artificial blood vessel and wound dressing.
6. e‒spun fibres
employed in a variety of applications such as filtration and thermal
insulation.
Applied Chemistry I: UNIT 2: Nanochemistry : Tag: Applied Chemistry : - Preparation of Nanomaterials
Applied Chemistry I: UNIT 2: Nanochemistry
Under Subject
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Related Subjects
English Essentials I
EN25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
தமிழர் மரபு - Heritage of Tamils
UC25H01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Calculus
MA25C01 Maths 1 M1 - 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I
PH25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Makerspace
ME25C04 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming C
CS25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming Python
CS25C02 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Fundamentals of Electrical and Electronics Engineering
EE25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Mechanical Engineering
ME25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Civil Engineering
CE25C01 1st Semester Civil Department | 2025 Regulation | 1st Semester 2025 Regulation
Essentials of Computing
CS25C03 1st Semester - AID CSE IT Department | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I Laboratory
PH25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Applied Chemistry I Laboratory
CY25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming C Laboratory
CS25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming Python Laboratory
CS25C02 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Engineering Drawing
ME25C01 EEE Mech Dept | 2025 Regulation | 2nd Semester 2025 Regulation
Basic Electronics and Electrical Engineering
EE25C04 1st Semester ECE Dept | 2025 Regulation | 2nd Semester 2025 Regulation

















