Applied Chemistry I: UNIT 3: Electrochemistry
Conductance (or) Conductivity of Electrolytes
Electrochemistry (Applied Chemistry) : Conductance (or) Conductivity of Electrolytes
CONDUCTANCE (OR) CONDUCTIVITY OF
ELECTROLYTES
1. Ohm's law
The resistance of a conductor
is directly proportional to its length and inversely proportional to its cross
sectional area
R ∝
l ……….. (1)
R ∝ l/a ……….. (2)
Combining (1) & (2)
R = ρ l/a
Where, R= Resistance in
ohms;
ρ = specific resistance;
l=
length in centimeter;
a=
area of cross‒section in cm2
Thus, if l = 1 cm and a = 1 cm2
2. Conductance (c)
The conductance of an
electrolyte is the reciprocal of its resistance
c = 1/R
Unit
of conductance:
c = 1/ohm = ohm‒1 (or) mho.
3. Specific resistance (ρ)
According to ohm's law
R ∝ l/a
R = ρ l/a (or) ρ = R × a/l
Where, ρ = specific
resistance
If l = 1 cm and a = 1 cm2,
then ρ = R
Thus, specific
resistance is defined as the resistance of a conductor, if its length is 1 cm
and area is 1 cm2.
Unit
of specific resistance:
ρ = [ ohm × cm2
] / cm = ohm.cm
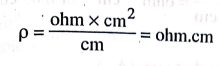
4. Specific Conductance (κ)
The reciprocal of
specific resistance is called specific conductance. (or) It is the conductance
of 1 cm3 of a material.
κ = 1/ρ
= 1/ [R × a/l]
κ = 1/R × l /a
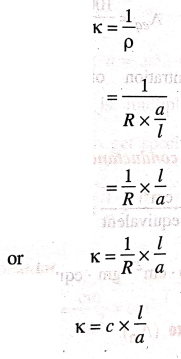
κ = c × l /a
Where, I/R=c
Unit
of Specific Conductance
κ= cm / [ohm × cm2]
κ= 1 / [ohm × cm] = ohm‒1
× cm‒1 or mho.cm‒1
5. Equivalent conductance (˄eq)
It is defined as the
conducting power of all the ions produced by dissolving 1 gram equivalent of an
electrolyte in the solution.
˄eq = 1000κ / C
Where, C =
concentration of the solution in gm equivalent weight.
Unit
of equivalent conductance
˄eq = [ cm3
/ gm.equivalent ] × mho.cm‒1
= mho.cm2 • gm
• equ‒1
6. Molar conductance
(˄m)
It is defined as
"the conducting power of all the ions produced by dissolving 1 mole of an
electrolyte in a given solution."
˄m
= 1000κ / C
Where, C= molar
concentration
Unit
of molar concentration
˄eq = cm3/mole × mho.cm‒1
= mho.cm2 •
mole‒1
7. Cell constant (x):
It is the ratio of
distance between two electrodes (l)
and area of the electrodes (a).
x = l/a
Where, l = length in cm; a = area in cm2
This cell constant is
multiplied with observed conductance (c) or (1/R) to get specific conductance (κ)
κ = 1/R × l/a
Unit of cell constant
x = cm
/ cm2 = cm‒1
1. Relationship between specific conductance (κ) and equivalent conductance (˄eq)
Imagine 1 cc (1 cm3)
of a solution of an electrolyte placed between two electrodes of 1 cm apart and
the cross‒sectional area of the electrodes is 1 cm2.
The conductance (C) of
the above solution will be its specific conductance (κ), because we having 1 cc
(1 cm3) of the electrolyte. Now,
Conductance (C) = κ
(Specific conductance).
Suppose 1 cc of the
solution contains 1 gm equivalent of the electrolyte, then the conductance of
the solution will be equal to its equivalent conductance (˄eq).
Conductance (C) = κ
(specific conductance)
= ˄eq (equivalent conductance).
Suppose the solution is
diluted to 1000 cc. Now, we have 1000 cc of the electrolyte. The conductance of
the solution is 1000 times of its specific conductance. But, even after the
dilution, the solution contains 1 gm equivalent of the electrolyte, so the conductance
measured will be its equivalent conductance.
Thus, the equivalent
conductance (˄eq) = 1000 × specific conductance
Λeq = 1000 × κ
If the solution is diluted
to V times, then
˄eq = V × κ
Where, V is the volume
of the solution in cc containing 1 gm equivalent of the electrolyte. If the
concentration is C gm equivalent per litre, then
˄eg = [1000 / C] × κ
2. Variation of specific and equivalent conductance on dilution
Specific conductance (κ)
of an electrolyte decreases on dilution. As the dilution increases, the
conducting power of 1 cc of the solution decreases because number of ions per
cc decreases due to diffusion.
Equivalent conductance
(˄eg) of an electrolyte increases on dilution. The conducting power of an
electrolyte depends on the number of ions and this increases with increasing
dilution, since the degree of ionisation of an electrolyte increases with
dilution.
Thus, the equivalent
conductance increases with increasing dilution whereas the specific conductance
decreases with increasing dilution.
3. Variation of equivalent conductance on dilution for strong and weak electrolytes
Equivalent conductance
of strong electrolytes like HCl, NaCl, etc., are always high and it slightly
increases with dilution. Strong electrolytes are completely ionized at infinite
dilution. The relationship between ˄ and concentration is linear for strong
electrolytes. So the equivalent conductance (˄0) for strong electrolytes
are obtained by extrapolating the line to zero concentration (Fig 3.1)
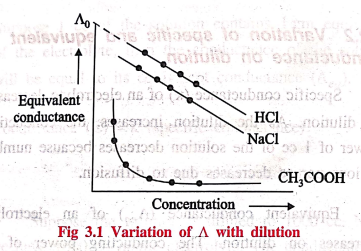
Equivalent conductance
of weak electrolytes like CH3COOH, NH4OH, etc., are very
low and it increases steadily with dilution. Even at infinite dilution weak electrolytes
are not completely ionised. The relationship between ˄ and concentration is not
linear for weak electrolytes. So the equivalent conductance cannot be obtained
by extrapolation.
4. Measurement of specific and equivalent conductance
Aim
The measurement of
electrical conductance of an electrolytic solution involves, the determination
of electrical resistance since the conductance is the reciprocal of electrical
resistance.
Procedure
The determination of
electrical resistance is carried out by using wheatstone bridge (Fig. 3.2.)
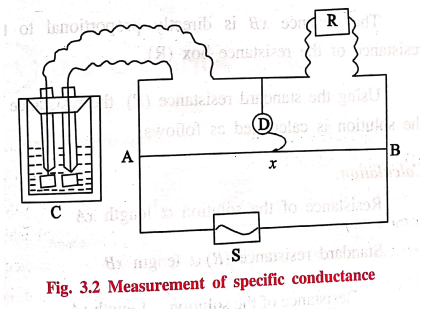
AB is a uniform wire
and a sliding contact x moves over
it. Alternate current is fed into the circuit through the source S. C is the
conductivity cell containing the solution of an electrolyte whose resistance is
to be measured. R is the resistance box and D is the head phone to detect the
current.
When current flows, a
known resistance R is introduced through the resistance box. The sliding
contact x is then moved along the
wire AB until a point of minimum sound (called null point) in the head phone is
detected. The lengths xA and xB can be measured from the scale fixed
below the wire AB.
The distance xA is directly proportional to the
resistance of the solution.
The distance xB is directly proportional to the
resistance of the resistance box (R).
Using the standard
resistance (R), the resistance of the solution is calculated as follows.
Calculation:
Resistance of the
solution α length xA
Standard resistance (R)
α length xB
Resistance of the
solution / Standard resistance (R) = Length xA
/ Length xB
Resistance of the
solution = [ Length xA / Length xA ] × R
(i) Reciprocal of this
resistance gives the conductance of the solution.
(ii) The value of
conductance is multiplied by cell constant to get the specific conductance (κ)
Specific conductance (κ) = conductance × cell
constant
= conductance × l/a
(iii) From the
normality of the solution and its specific conductance, equivalent conductance
can be calculated as follows
˄eq = (1000 / C) × κ
Applied Chemistry I: UNIT 3: Electrochemistry : Tag: Applied Chemistry : - Conductance (or) Conductivity of Electrolytes
Applied Chemistry I: UNIT 3: Electrochemistry
Under Subject
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Related Subjects
English Essentials I
EN25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
தமிழர் மரபு - Heritage of Tamils
UC25H01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Calculus
MA25C01 Maths 1 M1 - 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I
PH25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Makerspace
ME25C04 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming C
CS25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming Python
CS25C02 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Fundamentals of Electrical and Electronics Engineering
EE25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Mechanical Engineering
ME25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Civil Engineering
CE25C01 1st Semester Civil Department | 2025 Regulation | 1st Semester 2025 Regulation
Essentials of Computing
CS25C03 1st Semester - AID CSE IT Department | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I Laboratory
PH25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Applied Chemistry I Laboratory
CY25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming C Laboratory
CS25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming Python Laboratory
CS25C02 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Engineering Drawing
ME25C01 EEE Mech Dept | 2025 Regulation | 2nd Semester 2025 Regulation
Basic Electronics and Electrical Engineering
EE25C04 1st Semester ECE Dept | 2025 Regulation | 2nd Semester 2025 Regulation

















