Applied Chemistry I: UNIT 3: Electrochemistry
Electrode Potential
1. Origin of Electrode Potential 2. Illustration 3. Oxidation potential 4. Reduction potential 5. Nernst equation for electrode potential 6. Applications (or) Significance of Nernst equations (Electrode Potential) 7. Measurement of single electrode potential
ELECTRODE POTENTIAL
1. Origin of Electrode Potential
A metal (M) consists of
metal ions (Mn+) with valence electrons. When the metal (M) is
placed in a solution of its own salt, any one of the following reactions will
occur.
(i) Positive metal ions
may pass into the solution.
M → Mn+ + ne‒
(oxidation)
(ii) Positive metal
ions from the solution may deposit over the metal.
Mn+ + ne‒ → M (reduction)
The above reactions
indicate that the electrodes of a galvanic cell are at different potentials.
So, it is necessary to know how potential arises in an electrode.
2. Illustration
In order to understand
the origin of electrode potential, the following two examples are considered.
Example
1:
Zn electrodes dipped in ZnSO4 solution
When Zn electrode is
dipped in ZnSO4 solution, Zn goes into the solution as Zn2+
ions due to oxidation.
Zn → Zn2+ +
2e‒
Now, the Zn electrode
attains a negative charge, due to the accumulation of valence electrons on the
metal. The negative charges developed on the electrode attract the positive ions
from solution. Due to this attraction the positive ions remain close to the
metal. (Fig. 3.4.a)
Example
2:
Cu electrode dipped in CuSO4 solution
When Cu electrode is
dipped in CuSO4 solution, Cu2+ ions from the solution
deposit over the metal due to reduction.
Cu2+ + 2e‒
→ Cu
Now, the Cu electrode
attains a positive charge, due to the accumulation of Cu2+ ions on
the metal. The positive charges developed on the electrode attract the negative
ions from solution. Due to this attraction, the negative ions remain close to
the metal. (Fig. 3.4.b)

Thus, a sort of layer
(positive (or) negative ions) is formed all around the metal. This layer is
called Helmholtz electrical double layer.
This layer prevents further passing of the positive ions from or to the metal.
A difference of potential is consequently set up between the metal and the
solution. At equilibrium, the potential difference becomes a constant value,
which is known as the electrode potential of a metal.
Factors affecting electrode potential
The rate of the above
reactions depend on
(i) The nature of the
metal.
(ii) The temperature.
(iii) The concentration
of metal ions in solution.
Single electrode potential (E)
It is the measure of
tendency of a metallic electrode to lose or gain electrons, when it is in
contact with a solution of its own salt
Standard electrode potential (E°)
It is the measure of
tendency of a metallic electrode to lose or gain electrons, when it is in
contact with a solution of its own salt of 1 molar concentration at 25°C.
3. Oxidation potential
If oxidation occurs at
the electrode, at equilibrium, the potential of the electrode is oxidation
potential.
Zn ↔ Zn2+ +
2e‒
Thus, the tendency of
an electrode to lose electrons is called the oxidation potential.
4. Reduction potential
If reduction occurs at
the electrode, at equilibrium, the potential of the electrode is reduction
potential.
Cu2+ + 2e‒
↔ Cu
Thus, the tendency of
an electrode to gain electrons is called the reduction potential.
5. Nernst equation for electrode potential
Consider the following
redox reaction
Mn+ + ne‒
↔ M
For such a redox
reversible reaction, the free energy change (ΔG) and its equilibrium constant
(K) are inter related as M

where,
ΔG° = Standard free
energy change
The above equation (1)
is known as Van't Hoff isotherm.
The decrease in free
energy (‒ΔG) in the above reaction will produce electrical energy. In the cell,
if the reaction involves transfer of 'n' number of electrons, then 'n' faraday of electricity will flow. If
E is the emf of the cell, then the total electrical energy (nEF) produced in
the cell is
‒ΔG = nEF (or)
‒ΔG0 = nE0F
…………..(2)
where,
‒ΔG = decrease in free
energy change.
(or) ‒ ΔG°= decrease in
standard free energy change.
Comparing equation 1
and 2, it becomes
‒nEF = ‒nE°F + RT ln ( [M]/[Mn+]
) .......(3)
Dividing the above
equation (3) by ‒nF
[. the activity of solid metal [M] = 1]
E = E° ‒ RT/nF ln (1/ [Mn+]
)
In general, E = E° ‒
RT/nF ln ([Product]/ [Reactant] )
(or)
E = E° + RT/nF ln [Mn+]
(or)
E = E° + ( 2.303RT/nF)log[Mn+] ……………(4)

When, R= 8.314
J/K/mole; F = 96500 coulombs ;
T= 298 K (25°C), the
above equation becomes
E = E°red +
(0.0591/n)log[Mn+]
……………(5)

In general, E = E°red
+ (0.0591/n)logC
Similarly for oxidation
potential

E = E°oxi +
(0.0591/n)log[Mn+] …………(6)
The above equation
5&6 are known as "Nernst equation for single electrode
potential".
6. Applications (or) Significance of Nernst equations (Electrode Potential)
1. Nernst equation is
used to calculate electrode potential of unknown metal.
2. Corrosion tendency
of metals can be predicted.
3. It is used to
calculate the EMF of a cell.
4. Concentration of the
reactant can be calculated using the electrode potential.
5. Concentration of the
solution in galvanic cell can be determined.
6. pH of a solution can
be calculated by measuring the emf.
7. Measurement of single electrode potential
It is impossible to
determine the absolute value of a single electrode potential. But, we can
measure the potential difference between two electrodes potentiometrically, by
combining them to form a complete cell. For this purpose, 'reference electrode'
is used. Standard hydrogen electrode (SHE) is the commonly used reference
electrode, whose potential has been arbitrarily fixed as zero. The emf of the
cell is measured and it is equal to the potential of electrode.
It is very difficult to
set up a hydrogen electrode. So other electrodes called secondary reference
electrodes like calomel electrodes are used.
Example
Measurement of single electrode potential of Zn using saturated calomel electrode
To measure the
electrode potential of Zn, a cell is made by combining the saturated calomel
electrode with Zn electrode, the potential of which is to be determined (Fig.
3.5). Since the reduction potential of the coupled Zn electrode is less than E°
of calomel electrode (+0.2422 V), the calomel electrode will act as cathode and
zinc electrode will act as anode.

The following cell reaction
will occur in the above cell
At anode: Zn ↔ Zn2+
+ 2e‒ (oxidation)
At cathode: Hg2Cl2(s)
+ 2e‒ ↔ 2Hg(l) + 2Cl‒ (reduction)
Cell reaction: Zn + Hg2Cl2 (s)
↔ ZnCl2 + 2Hg(l)
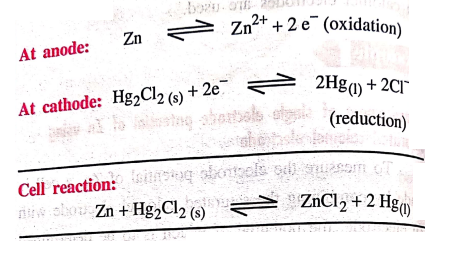
The emf of the above
cell is measured and the electrode potential is calculated from the emf as
follows
Ecell = E°right
‒ E°left
Ecell = E°cal
‒ E°Zn
E°Zn = E°cal
‒ Ecell
= +0.2422 ‒ 1.0025
E°Zn = ‒0.7603
volt.
ie., the reduction
potential of Zn electrode = ‒0.7603 volt.
Applied Chemistry I: UNIT 3: Electrochemistry : Tag: Applied Chemistry : - Electrode Potential
Applied Chemistry I: UNIT 3: Electrochemistry
Under Subject
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Related Subjects
English Essentials I
EN25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
தமிழர் மரபு - Heritage of Tamils
UC25H01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Calculus
MA25C01 Maths 1 M1 - 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I
PH25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Makerspace
ME25C04 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming C
CS25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming Python
CS25C02 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Fundamentals of Electrical and Electronics Engineering
EE25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Mechanical Engineering
ME25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Civil Engineering
CE25C01 1st Semester Civil Department | 2025 Regulation | 1st Semester 2025 Regulation
Essentials of Computing
CS25C03 1st Semester - AID CSE IT Department | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I Laboratory
PH25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Applied Chemistry I Laboratory
CY25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming C Laboratory
CS25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming Python Laboratory
CS25C02 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Engineering Drawing
ME25C01 EEE Mech Dept | 2025 Regulation | 2nd Semester 2025 Regulation
Basic Electronics and Electrical Engineering
EE25C04 1st Semester ECE Dept | 2025 Regulation | 2nd Semester 2025 Regulation

















