Applied Chemistry I: UNIT 3: Electrochemistry
Electrochemical Cells (or) Galvanic Cells
Galvanic cells are electrochemical cells in which the electrons, transferred due to redox reaction, are converted into electrical energy.
ELECTROCHEMICAL CELLS OR GALVANIC
CELLS
Galvanic cells are
electrochemical cells in which the electrons, transferred due to redox
reaction, are converted into electrical energy.
Cell device (Construction)
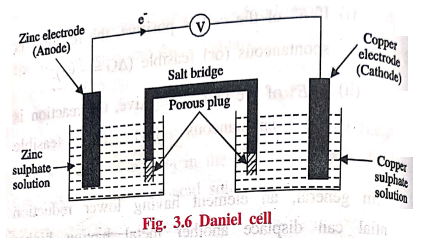
Galvanic cell consists
of a zinc electrode dipped in 1 M ZnSO4 solution and a copper
electrode dipped in 1 M CuSO4 solution. Each electrode is known as a
half cell. The two solutions are inter connected by a salt bridge and the two
electrodes are connected by a wire through a voltmeter.
Reactions occurring in the cell
At
anode: Oxidation takes place in the zinc electrode by the
liberation of electrons, so this electrode is called negative electrode or
anode.
At
cathode: Reduction takes place in the copper electrode by
the acceptance of electrons, so this electrode is called the positive electrode
or cathode.

The electrons liberated
by the oxidation reaction flow through the external wire and are consumed by
the copper ions at the cathode.
Salt bridge
It consists of a U‒tube
containing saturated solution of KCl or NH4NO3 in agar‒agar
gel. It connects the two half cells of the galvanic cells.
Functions of salt bridge
(i) It eliminates
liquid junction potential.
(ii) It provides the
electrical continuity between the two half cells.
Conditions for a cell to act as standard cell
The conditions for an
electrochemical cell to act as a standard cell are
(i) The e.m.f of the
cell is reproductive.
(ii) The temperature‒coefficient
of e.m.f (change in e.m.f with temperature) should be very low.
1. Representation of a galvanic cell (or) Cell diagram
(i) A galvanic cell consists
of two electrodes anode and cathode.
(ii) The anode is
written on the left hand side while the cathode is written on the right hand
side.
(iii) The anode must be
written by writing electrode metal first and then electrolyte. These two are
separated by a vertical line or a semicolon. The electrolyte may be written by
the formula of the compound (or) by ionic species.
Examples
(a) Zn/Zn2+
(or) Zn/ZnSO4 (or) Zn; Zn2+
(b) Standard hydrogen
electrode
Pt, H2 (1 atm); H+ (1 M)
(iv) The cathode must
be written by writing electrolyte first and then the electrode metal. These two
are separated by a vertical line or a semicolon.
Example: Cu2+/Cu
(or) CuSO4/Cu (or) Cu2+; Cu
(v) The two half cells
are separated by a salt bridge, to which is indicated by two vertical lines.
Using the above
representation, the galvanic cell is represented as follows.
Zn / ZnSO4 (1 M) // CuSO4
(1 M) / Cu
(or) Zn/Zn2+ (1 M) // Cu2+
(1 M)/Cu
(or) Zn; Zn2+ (1 M) // Cu2+
(1 M); Cu
Applied Chemistry I: UNIT 3: Electrochemistry : Tag: Applied Chemistry : - Electrochemical Cells (or) Galvanic Cells
Applied Chemistry I: UNIT 3: Electrochemistry
Under Subject
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Related Subjects
English Essentials I
EN25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
தமிழர் மரபு - Heritage of Tamils
UC25H01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Calculus
MA25C01 Maths 1 M1 - 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I
PH25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Makerspace
ME25C04 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming C
CS25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming Python
CS25C02 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Fundamentals of Electrical and Electronics Engineering
EE25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Mechanical Engineering
ME25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Civil Engineering
CE25C01 1st Semester Civil Department | 2025 Regulation | 1st Semester 2025 Regulation
Essentials of Computing
CS25C03 1st Semester - AID CSE IT Department | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I Laboratory
PH25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Applied Chemistry I Laboratory
CY25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming C Laboratory
CS25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming Python Laboratory
CS25C02 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Engineering Drawing
ME25C01 EEE Mech Dept | 2025 Regulation | 2nd Semester 2025 Regulation
Basic Electronics and Electrical Engineering
EE25C04 1st Semester ECE Dept | 2025 Regulation | 2nd Semester 2025 Regulation

















