Applied Physics I: Chapter 2: Properties of Matter - Viscosity and Surface Tension
Viscosity and Surface Tension: Important part-A 2 marks Short Questions and Answers
Properties of Matter - Viscosity and Surface Tension (Applied Physics I) - Important part-A 2 marks Short Questions and Answers
ANNA
UNIVERSITY PART 'A' QUESTIONS & ANSWERS
1) Define critical
velocity of a fluid.
The
velocity at which the steady, streamline flow changes into turbulent flow is
called critical velocity.
2) Define streamline
flow.
Streamline
Flow
In
a streamline flow of a fluid, the velocity of the fluid does not change with
time, either in magnitude or in direction.
3) Define Coefficient
of viscosity.
The
Coefficient of viscosity is defined as the tangential force per unit area
required to maintain a unit velocity gradient.
Tangential
Force F ∝ surface area of the
layer (A)
F
∝ Velocity gradient
F ∝
A
dv/dx
(or)
F = ‒ηA dv/dx pascal second
where
η is the Coefficient of viscosity. The negative sign indicates that force is
acting opposite to the direction of viscosity.
4) Differentiate
between streamline flow and turbulent flow.

Streamline
flow
1.
A stream line flow is the flow in which each particle in the liquid moves in
the same path and has same velocity as that of that of its preceding particle.
2.
The process is regular.
3.
The velocity at every point within the liquid remains constant both in
magnitude and direction and it does not exceed the critical velocity.
4.
It does not cause eddies or whirlpools
5.
Example: Flow of liquid through a capillary tube.
Turbulent
flow
1.
A turbulent flow is the flow in which the particles moves in different
directions and with different velocities.
2.
The process is irregular.
3.
The velocity is not constant and it exceeds the critical velocity.
4.
It causes eddies and whirlpools.
5.
Example: Flow of liquid from a tap.
ADDITIONAL
PART 'A' QUESTIONS
1) Define viscosity.
Viscosity
or internal friction of the liquid is the property of a liquid by virtue of
which the liquid opposes the relative motion between its different layers.
2) Give the examples
for high viscous and low viscous liquids.
High
viscous liquids: Thick liquids such as castor oil, honey, coal tar, glycine
etc.,
Low
viscous liquids: Thin liquids such as water, alcohol, spirit, kerosine oil etc.
3) Mention some
practical applications of the knowledge of viscosity.
(i)
Lubrication: The phenomenon of viscosity and its variation with temperature
helps to use a suitable lubricant for a certain machine. Liquids with moderate
viscosities are good lubricants for light machinery. High viscous oils are used
as lubricants for heavy machinery.
(ii)
Damping of instruments: The viscosity of air and some liquids are used for
damping the motion of some instruments. Paddles or vanes of such instruments
move in the liquid or air. The viscous force opposes the motion. Thus the
moving parts of the instruments attain the final position quickly.
4) Which rain drops
fall faster, bigger one or smaller one? why?
Velocity
v = P/4ηl (r2‒x2)
∴ v ∝ r2
Since
the velocity is directly proportional to the radius of the droplet, bigger rain
drops fall faster than the smaller one.
5) Why do we find fine
cloud particles floating in the sky?
Cloud
is an assembly of a large number of small droplets which are formed by the
condensation of water vapour on dust or small particles. Under the action of
gravity, upthrust of air and viscous drag of air takes place, the downward
terminal velocity 'v' of the droplet is given by
v
= P/4ηl (r2‒x2)
∴ v ∝ r2
As
the radius of the rain droplets present in the cloud is very small, the
terminal velocity is also very small. Hence, cloud particles appear floating.
6) What will happen if
the density of the medium is greater than that of the body?
If
the density of the medium is greater than the density of the body, the terminal
velocity becomes negative and the sphere moves upwards with a constant
velocity. Slow bubbling out of CO2 from a soda water bottle is an
example of this phenomenon.
7) Define critical
velocity.
The
velocity at which the streamline flow is changed into turbulent flow is called
critical velocity.
8) Is it possible to
determine the viscosity of water by Stokes' method?
Stokes'
method is applicable for determining the coefficient of viscosity of highly
viscous liquids like castor oil. In the case of water (medium), there is slip
between the body and medium. Hence this method is not suitable for liquids like
water (or) less denser liquids.
9) Mention some
practical applications of the knowledge of viscosity.
(i)
Lubrication: The knowledge of viscosity and its variation with temperature
helps to identity a suitable lubricant for a certain machine. Liquids with
moderate viscosities are good lubrications for light machinery. High viscous
oils are used as lubrications for heavy machinery.
(ii)
Damping of instruments: The viscosity of air and some liquids are used for
dampling the motion of some instruments. The viscous force opposes the motion.
Thus the moving parts of the instruments attain the final position quickly.
10) Which rain drops
fall faster, big ones or small ones? Why?
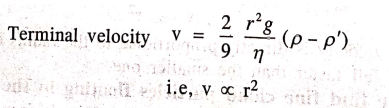
Terminal
velocity v = 2/9 . r2g/η . (ρ‒ρ')
∴ i.e, v ∝ r2
Since
the terminal velocity is directly proportional to the square of the radius of
the rain drop, bigger rain drops fall faster than smaller ones.
11) What is the
principle used in stokes' method to determine the viscosity of a high viscous
liquid.
When
a spherical object, like steel ball moves through a high viscous liquid, when
the viscous force is equal to the buoyout force it attains the terminal
velocity. By finding the termial velocity, the co‒efficient of viscosity shall
be determined.
12. List any four
applications of Stokes' law.
(i)
Stokes' law/principle is used in understanding the phenomenon of settling of
particles in liquids.
(ii)
This method is used to find the viscocity of high viscous liquids.
(iii)
Stokes' law is used in various fields such as sedimentation, Aerosol science
etc.
(iv)
This principle is also used in fluid dynamics, parachute designing etc.
13. Define Surface
tension.
Definition
Surface
tension (σ) is defined as the force per unit length acting along the surface of
a liquid. It is measured in units of force per unit length (N/m).
The
formula to calculate surface tension is
σ
= F/L
Where,
•
σ is the surface tension,
•
F is the force acting along the surface, and
•
L is the length over which the force acts.
14. Brief the principle
used in drop weight method for determing the surface tension of the liquid.
This
method is based on the principle that the weight of a drop falling slowly out
of a capillary held vertically is directly proportional to its surface tension.
The drop falls when its weight just exceeds surface tension.
15. It is easier to
spray water when soap is added to it than when is is pure. Why?
Yes,
when soap is added to water, the surface tension decreases. Therefore less work
is required to spray water. Hence, it is easier to spray soap water than pure
water.
16. In the case of
mercury, there is capillary depression. Why?
In
mercury there exist a capillary depression, because the cohesive forces in the
liquid molecules are greater than the forces of attraction between the glass
and liquid molecules.
17. Water wets the
glass surface while mercury does not. why?
Water
wets the glass surface, because the forces of adhesion between water and glass
are greater than the forces of cohesion between water molecules.
18. If two bubbles of
different size are blown at opposite ends of a pipe, what will happen?
If
two bubbles of different sizes are blown at opposite ends of a pipe, then, the
smaller bubble will contract and the larger bubble will expand, showing that
the air flows from smaller bubble which has less pressure.
19. List any four
applications of drop weight method.
(i)
Drop weight method is used to determine the purity of liquids in pharmaceutical
and chemical industries.
(ii)
This method is used to check the quality of paints, lubricants, inks,
beverages, etc.,
(iii)
It is mainly used in formulation of detergents and surfactants.
(iv)
This method is also used to study the effects of temperature and additives.
Applied Physics I: Chapter 2: Properties of Matter - Viscosity and Surface Tension : Tag: Applied Physics : - Viscosity and Surface Tension: Important part-A 2 marks Short Questions and Answers
Applied Physics I: Chapter 2: Properties of Matter - Viscosity and Surface Tension
Under Subject
Applied Physics I
PH25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Related Subjects
English Essentials I
EN25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
தமிழர் மரபு - Heritage of Tamils
UC25H01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Calculus
MA25C01 Maths 1 M1 - 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I
PH25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Makerspace
ME25C04 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming C
CS25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming Python
CS25C02 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Fundamentals of Electrical and Electronics Engineering
EE25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Mechanical Engineering
ME25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Civil Engineering
CE25C01 1st Semester Civil Department | 2025 Regulation | 1st Semester 2025 Regulation
Essentials of Computing
CS25C03 1st Semester - AID CSE IT Department | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I Laboratory
PH25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Applied Chemistry I Laboratory
CY25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming C Laboratory
CS25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming Python Laboratory
CS25C02 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Engineering Drawing
ME25C01 EEE Mech Dept | 2025 Regulation | 2nd Semester 2025 Regulation
Basic Electronics and Electrical Engineering
EE25C04 1st Semester ECE Dept | 2025 Regulation | 2nd Semester 2025 Regulation

















