Applied Chemistry I: UNIT 1: Water Technology
Remediation (or) Treatment of Industrial Feed Water (Softening or Conditioning Methods)
Softening of water can be done in two methods 1. Internal treatment. 2. External treatment.
REMEDIATION (OR) TREATMENT OF INDUSTRIAL
FEED WATER (SOFTENING OR CONDITIONING METHODS)
Water used for
industrial purposes should be free from hardness producing substances,
suspended impurities and dissolved gases etc. The process of removing hardness
producing salts from water is known as softening (or) conditioning of water.
Softening of water can
be done in two methods
1. Internal treatment.
2. External treatment.
1. Internal Conditioning (or) Internal Treatment (or) Boiler Compounds
It involves the removal
of scale forming substance, which were not completely removed in the external
treatment, by adding chemicals directly into the boiler. This chemicals are
also called boiler compounds.
1. Phosphate conditioning
Scale formation can be
avoided by adding sodium phosphate. It is used in high pressure boilers. The
phosphate reacts with Ca2+ and Mg2+ salts to give soft
sludges of calcium and magnesium phosphates.
3CaSO4 + 2Na3PO4
→ Ca3(PO4)2 + 3Na2SO4
Generally 3 types of
phosphates are employed.
(a)
Trisodium phosphate ‒ Na3PO4 (Too
alkaline) ‒ used for too acidic water.
(b)
Disodium hydrogen phosphate ‒ Na2HPO4
(weakly alkaline) used for weakly acidic water.
(c)
Sodium dihydrogen phosphate ‒ NaH2PO4
(acidic) used for alkaline water.
2. Colloidal conditioning
Scale formation can be
avoided by adding colloidal conditioning agents like kerosene, agar‒agar,
gelatin, etc., It is used in low pressure boilers. These colloidal substances
get coated over the scale forming particles and convert them into non‒adherent,
loose precipitate called sludge, which can be removed by blow down operation.
3. Sodium aluminate conditioning
Sodium aluminate (NaAlO2)
under goes hydrolysis in boiler water to give gelatinous white precipitate of
aluminium hydroxide and sodium hydroxide.
NaAlO2 + 2H2O
→ Al(OH)3 + NaOH
The sodium hydroxide,
thus formed, precipitates magnesium as magnesium hydroxide. The gelatinous precipitates
of aluminium hydroxide and magnesium
hydroxide entrap the
colloidal silica and finely divided solids and settled easily. This can be
removed easily by blow down operations.
4. Calgon conditioning
Calgon is sodium hexa
meta phosphate Na2 [Na4(PO3)6].
This substance interacts with calcium ions forming a highly soluble complex and
thus prevents the precipitation of scale forming salt.
2CaSO4 + Na2[Na4(PO3)6]
→ Na2[Ca2(PO3)6] + 2Na2SO4.
The complex Na2[Ca2(PO3)6]
is soluble in water and there is no problem of sludge disposal. So calgon
conditioning is better than phosphate conditioning.
2. External conditioning (or) External Treatments
It involves the removal
of hardness producing salts from the water before feeding into the boiler. The
external treatment can be done by two process
1. Demineralisation
(or) Ion‒exchange process
2. Zeolite process.
1. Ion Exchange (or) Demineralisation process
This process removes
almost all the ions (both anions and cations) present in hard water.
The soft water,
produced by zeolite processes, does not contain hardness producing Ca2+
and Mg2+ ions, but it contains other ions like Na+, K+,
SO42‒, Cl‒ etc., On the other hand D.M.
(Demineralised) water does not contain both anions and cations.
Thus, a soft water is
not demineralised water whereas a demineralised water is soft
Resins
Demineralisation
process is carried out by using ion exchange resins, which are long chain,
cross linked, insoluble organic polymers with a microporous structure.
The functional groups
attached to the chains are responsible for the ion exchanging properties. The
following two types of ion exchange resins are used.
1. Cation exchange
resin (or) cation exchanger
2. Anion exchange resin
(or) Anion exchanger
1. Cation exchanger
Resins containing
acidic functional groups (‒COOH, ‒SO3H) are capable of exchanging
their H+ ions with other cations of hard water. Cation exchange resin
is represented as RH2.
Examples
(i) Sulphonated coals.
(ii) Sulphonated
polystyrene.
R‒SO3H; R‒COOH
≡ RH2
2. Anion Exchanger
Resins containing basic
functional groups (‒NH2,‒OH) are capable of exchanging their anions with
other anions of hard water. Anion exchange resin is represented as R (OH)2.
Examples
(i) Cross‒linked
quaternary ammonium salts.
(ii) Urea‒formaldehyde
resin.
R‒NR3OH; R‒OH;
R‒NH2 ≡ R(OH)2
Process
The hard water first
passed through a cation exchange column, (Fig. 1.3) which absorbs all the
cations like Ca2+, Mg2+, Na+, K+,
etc., present in the hard water.
RH2 + CaCl2
→ RCa + 2HCI
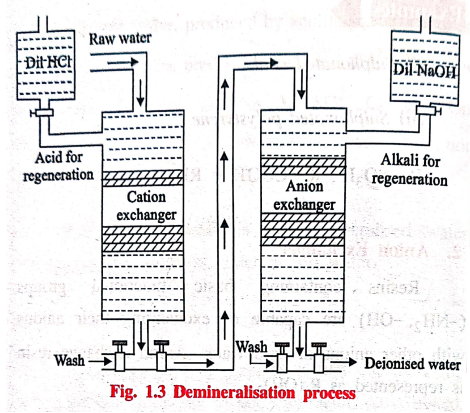
RH2 + MgSO4
→ RMg + H2SO4
RH+ NaCl → RNa + HCI
The cation free water
is then passed through a anion exchange column, which absorbs all the anions
like Cl‒, SO42‒, HCO3‒,
etc., present in the water.
R’(OH)2 +
2HC1 → R'Cl2 + 2H2O
R'(OH)2 + H2SO4
→R'SO4 + 2H2O
The water coming out of
the anion exchanger is completely free from cations and anions. This water is
known as demineralized water (or) deionised water.
Regeneration
When the cation
exchange resin is exhausted, it can be regenerated by passing a solution of dil
HCl (or) dil H2SO4.
RCa + 2HCl → RH2
+ CaCl2
RNa + HCl → RH + NaCl
Similarly, when the
anion exchange resin is exhausted, it can be regenerated by passing a solution
of dil NaOH.
R'Cl2 +
2NaOH → R'(OH)2 + 2NaCl.
Advantages of ion‒exchange process over Zeolite process
(i) Highly acidic (or)
alkaline water can be treated by this process.
(ii) The water obtained
by this process will have very low hardness (nearly 2 ppm).
(iii) In addition to Ca2+
and Mg2+, it removes almost all the ions (Na+, K+,
SO42‒, Cl‒)
Disadvantages of ion‒exchange process
(i) Water containing
turbidity, Fe and Mn cannot be treated, because turbidity reduces the output
and Fe, Mn form stable compound with the resin.
(ii) The equipment is
costly and more expensive chemicals are needed.
2. Zeolite (or) Permutit process
Zeolites are naturally
occurring hydrated sodium aluminosilicate. Its general formula is
Na2O. Al2O3.
xSiO2. yH2O. (x=2−10, y = 2 − 6).
(i)
Natural zeolites
These are green sand
and non‒porous.
(ii)
Synthetic Zeolites
These are known as
permutits. Zeolites are porous and possess gel like structure, hence it is
generally used for water softening.
Synthetic zeolite is
represented by Na2Ze. The sodium ions which are loosely held in Na2Ze
are replaced by Ca2+ and Mg2+ ions present in the water.
Process
When hard water is
passed through a bed of sodium zeolite (Na2Ze), kept in a cylinder
(Fig. 1.4), it exchanges its sodium ions with Ca2+ and Mg2+
ions present in the hard water to form calcium and magnesium zeolites. The
various reactions taking place during softening process are
Ca(HCO3)2
+ Na2Ze → CaZe + 2NaHCO3

Mg(HCO3)2
+ Na2Ze → MgZe + 2NaHCO3
CaSO4 + Na2Ze
→ CaZe + Na2SO4
MgSO4 + Na2Ze
→ MgZe + Na2SO4
CaCl2 + Na2Ze
→ CaZe + 2NaCl
MgCl2 + Na2Ze
→ MgZe + 2NaC1
The softened water is
enriched with large amount of sodium salts, which do not cause any hardness,
but cannot be used in boilers.
Regeneration
After some time zeolite
gets exhausted. The exhausted zeolite is again regenerated by treating with 10%
solution of NaCl.
CaZe +2NaCl → Na2Ze
+ CaCl2
MgZe + 2NaCl → Na2Ze
+ MgCl2
Advantages of Zeolite process
(i) Water obtained by
this process will have only hardness of 1‒2 ppm.
(ii) This method is
cheap, because the regenerated zeolite can be used again.
(iii) No sludge is
formed during this process.
(iv) The equipment used
is compact and occupies a small space.
(v) Its operation is
easy.
Disadvantages of Zeolite process
(i) Turbid water cannot
be treated, because it blocks the pores of the zeolite bed.
(ii) Acidic water
cannot be treated, because it decomposes the structure of zeolite.
(iii) The softened
water contains more dissolved sodium salts like NaHCO3, Na2CO3,
etc. When such water is boiled in boilers, CO2 and NaOH is produced
resulting in boiler corrosion and caustic embrittlement.
(iv) Water containing
Fe, Mn cannot be treated, because regeneration is very difficult.
(v) This process cannot
be used for softening brackish water, because brackish water contains Na+
ions. So the ion exchange reaction will not occur.
Table 1.3 Differences
between Zeolite and Demineralisation process

Zeolite process
1. It exchanges only cations.
2. Acidic water cannot
be treated because acid decomposes the zeolite.
3. The treated water contains
relatively large amount of dissolved salts, which leads to priming, foaming and
caustic embrittlement in boilers.
4. Water containing turbidity,
Fe, Mn cannot be treated.
Demineralisation process
1. It exchanges cations
as well as anions.
2. Acidic water can be
treated.
3. The treated water
does not contain any dissolved salts. Hence there is no priming and foaming.
4. Here also water containing
Fe, Mn cannot be treated.
Applied Chemistry I: UNIT 1: Water Technology : Tag: Applied Chemistry : - Remediation (or) Treatment of Industrial Feed Water (Softening or Conditioning Methods)
Applied Chemistry I: UNIT 1: Water Technology
Under Subject
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Related Subjects
English Essentials I
EN25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
தமிழர் மரபு - Heritage of Tamils
UC25H01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Calculus
MA25C01 Maths 1 M1 - 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I
PH25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Makerspace
ME25C04 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming C
CS25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming Python
CS25C02 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Fundamentals of Electrical and Electronics Engineering
EE25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Mechanical Engineering
ME25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Civil Engineering
CE25C01 1st Semester Civil Department | 2025 Regulation | 1st Semester 2025 Regulation
Essentials of Computing
CS25C03 1st Semester - AID CSE IT Department | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I Laboratory
PH25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Applied Chemistry I Laboratory
CY25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming C Laboratory
CS25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming Python Laboratory
CS25C02 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Engineering Drawing
ME25C01 EEE Mech Dept | 2025 Regulation | 2nd Semester 2025 Regulation
Basic Electronics and Electrical Engineering
EE25C04 1st Semester ECE Dept | 2025 Regulation | 2nd Semester 2025 Regulation

















