Applied Chemistry I: UNIT 1: Water Technology
Treatment of Water for Domestic Supply (or) Municipal Water Treatment
Rivers and lakes are the most common sources of water used by municipalities.
TREATMENT OF WATER FOR DOMESTIC
SUPPLY (OR) MUNICIPAL WATER TREATMENT
Rivers and lakes are
the most common sources of water used by municipalities. These water should be
free from colloidal impurities, domestic sewages, industrial effluents and
disease producing bacterias. Hence domestic supply of water involves the
following stages in the purification processes.

1. Primary Treatment
1. Screening
It is a process of
removing the floating materials like, leaves, wood pieces, etc. from water. The
raw water is through a screen, having large number of holes, which retains the
floating materials and allows the water to pass.
2. Aeration
The process of mixing
water with air is known as aeration. The main purpose of aeration is
(i) to remove gases
like CO2, H2S and other volatile stat impurities causing
bad taste and odour to water.
(ii) to remove ferrous
and manganous salts as insoluble ferric and manganic salts.
(iii) Promotes taste
and odour of water.
(iv) Increases the
oxygen concentration in water.
3. Sedimentation
It is a process of
removing suspended impurities by allowing stand undisturbed for 2‒6 hours the
water to in a big tank. Most of the suspended particles settle down due to
forces of gravity, and they are removed. Sedimentation removes only 75% of the
suspended impurities.
4. Coagulation
Finely divided clay,
silica, etc. do not settle down easily and hence cannot be removed by
sedimentation. Such impurities are removed by coagulation method.
In this method certain
chemicals, called coagulants, like alum, Al2(SO4)3
etc., are added to water. When the Al2(SO4)3
is added to water, it gets hydrolysed to form a gelatinous precipitate of
Al(OH)3. The gelatinous precipitate of Al(OH)3 entraps
the finely divided and colloidal impurities, settles to the bottom and can be
removed easily.
5. Filtration
It is the process of
removing bacteria, colour, taste, odour and suspended particles, etc., by
passing the water through filter beds containing fine sand, coarse sand and
gravel. A typical sand filter is shown in the figure 1.5.
The sand filter
consists of a tank containing a thick top layer of fine sand followed by coarse
sand, fine gravel and coarse gravel. When the water passes through the
filtering medium, it flows through the various beds slowly. The rate of
filtration decreases slowly due to the clogging of impurities in the pores of
the sand bed. When the rate of filtration becomes very slow, the filtration is
stopped and the thick top layer of fine sand is scrapped off and replaced with
clean sand. Bacterias are also partly removed by this process.

II. Sterilisation (or) Disinfection
The process of
destroying the harmful bacterias is known as sterilisation or disinfection. The
chemicals used for this purpose are called disinfectants. This process can be
carried out by the following methods.
1. By using ozone
Ozone is a powerful
disinfectant and is readily absorbed by water. Ozone is highly unstable and
breaks down to give nascent oxygen.
O3 → O2
+ [O]
The nascent oxygen is a
powerful oxidising agent and kills the bacterias.
Advantages
(a) O3 is a
multi functional agent. It acts as a bleaching, sterlizing, deodourising agent.
(b) It is harmless in
drinking water.
Disadvantages
(a) This process is
costly and cannot be used in large scale.
(b) Ozone is unstable
and cannot be stored for long time.
2. By using ultraviolet (UV) radiations
UV rays are produced by
passing electric current through mercury vapour lamp. This is particularly
useful for sterilizing water in swimming pool.
Advantages
(i) No requirements of
any chemical.
(ii) No changes in
colour and odour.
(iii) It is a very good
cleaning agent.
Disadvantages
(i) It is costly.
(ii) Turbid water
cannot be treated.
3. By chlorination
The process of adding
chlorine to water is called chlorination. Chlorination can be done by the
following methods.
(a) By adding chlorine gas
Chlorine gas can be
bubbled in the water as a very good disinfectant.
Advantages
(i) Chlorine gas is
harmless.
(ii) It is highly
effective and economical.
Disadvantages
(i) Handling is
difficult.
(ii) Excess
chlorination causes unpleasant odour.
(b) By adding chloramine
When chlorine and
ammonia are mixed in the ratio 2:1, a compound chloramine is formed.
Cl2 + NH3
→ CINH2 (Chloramine) + HCI
CINH2 + H2O
→ NH3 + HOCI (Hypochlorous acid)
Chloramine compounds
decompose slowly to give chlorine. It is a better disinfectant than chlorine.
Advantages
(i) It reduces the
formation of cancer causing disinfection by products.
(ii) It is more stable
and thus provides longer lasting residual disinfectant action.
Disadvantages
(i) Formation of
dichloramine and trichloramine affect the taste and odour of water.
(ii) It can cause
respiratory problems.
(c) By adding bleaching powder
When bleaching powder
is added to water, it produces hypochlorous acid (HOCl). HOCl is a powerful germicide.
CaOCl2 (Bleaching
powder) + H2O → Ca(OH)2 + Cl2
Cl2 + H2O
→ HCI + HOCI (Hypochlorous acid)
HOCI + Bacterias → Bacterias
are killed
Advantages
(i) It is eco‒friendly and effective
disinfectant.
(ii) It has long shelf life.
Disadvantages
(i) Irritation to skin
and respiratory system.
(ii) Harmful to aquatic
life.
(iii) Decomposes
overtime.
Break point chlorination
Chlorine may be added
to water directly as a gas (or) in the form of chloramine (or) bleaching
powder.
During this process the
following impurities, present in the water, are destroyed.
(i) Bacteria
(ii) Organic impurities
(iii) Reducing
substances (Fe2+, H2S etc.,)
(iv) Free ammonia
When chlorine is
applied to water, the results obtained can be depicted graphically in the
following figure 1.6. The graph is drawn between the amount of chlorine, added
to water, and the residual chlorine. It is explained as follows.
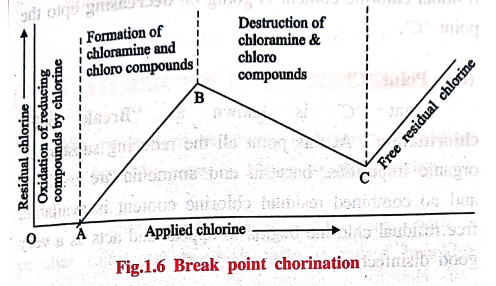
Explanation of the graph
The graph is explained
based on the curves and points.
(i)
Curve OA
The curve
"OA" shows, the applied chlorine gas is utilized for the oxidation of
reducing substances present in the water. So upto point "A" no Cl2
gas is available for disinfection.
(ii)
Curve AB
The curve
"AB" shows the formation of chloramine and chloro compound. As the
amount of applied chlorine increases the amount of combined residual chlorine
also increases.
(iii)
Curve BC
The curve
"BC" shows the destruction of chloramine and chloro compounds, where
the combined residual chlorine content is going on decreasing upto the point
'C'.
(iv)
Point 'C'
Point 'C' is known as
"Break point chlorination". At this point all the reducing
substances, organic impurities, bacteria and ammonia are oxidized and no
combined residual chlorine content is available, free residual chlorine begins
to appear and acts as a very good disinfectant.
(v)
Curve CD
The curve
"CD" shows the free residual chlorine gas is going on increasing as
the applied chlorine gas increases. It shows that the sterilization effect is
very good.
Break point chlorination definition
It is the point where
all the bacterias, reducing substances, organic compounds and ammonia are
completely removed and the applied chlorine begins to act as a powerful
disinfectant.
Advantages of break point chlorination
1. It completely
removes colour, odour and bad taste from water.
2. It also destroys the
bacterias.
3. It oxidizes all the
organic matters and other reducing substances like Fe2+, Mn2+,
H2S etc.,
Applied Chemistry I: UNIT 1: Water Technology : Tag: Applied Chemistry : - Treatment of Water for Domestic Supply (or) Municipal Water Treatment
Applied Chemistry I: UNIT 1: Water Technology
Under Subject
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Related Subjects
English Essentials I
EN25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
தமிழர் மரபு - Heritage of Tamils
UC25H01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Calculus
MA25C01 Maths 1 M1 - 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I
PH25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Makerspace
ME25C04 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming C
CS25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming Python
CS25C02 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Fundamentals of Electrical and Electronics Engineering
EE25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Mechanical Engineering
ME25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Civil Engineering
CE25C01 1st Semester Civil Department | 2025 Regulation | 1st Semester 2025 Regulation
Essentials of Computing
CS25C03 1st Semester - AID CSE IT Department | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I Laboratory
PH25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Applied Chemistry I Laboratory
CY25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming C Laboratory
CS25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming Python Laboratory
CS25C02 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Engineering Drawing
ME25C01 EEE Mech Dept | 2025 Regulation | 2nd Semester 2025 Regulation
Basic Electronics and Electrical Engineering
EE25C04 1st Semester ECE Dept | 2025 Regulation | 2nd Semester 2025 Regulation

















