Applied Chemistry I: UNIT 1: Water Technology
Troubles of Industrial Feed Water Containing Impurities (hardness) (or) Boiler Troubles
If hard water obtained from natural sources is fed directly into the boilers, the following troubles may arise. 1. Formation of Scales and sludges in boilers. 2. Priming and foaming (carry over). 3. Caustic embrittlement. 4. Boiler corrosion.
TROUBLES OF INDUSTRIAL FEED WATER
CONTAINING IMPURITIES (HARDNESS) (OR) BOILER TROUBLES
If hard water obtained
from natural sources is fed directly into the boilers, the following troubles
may arise.
1. Formation of Scales
and sludges in boilers.
2. Priming and foaming
(carry over).
3. Caustic
embrittlement.
4. Boiler corrosion.
1. Formation of Scales and Sludges in boilers
When water is
continuously converted into steam in boilers (or) heat exchangers, the
concentration of dissolved salts in water increases progressively. When the
concentration of the salts reaches their saturation point, they are thrown out
in the form of precipitates on the inner walls of the boilers (or) heat
exchangers. The least soluble one gets precipitated first.
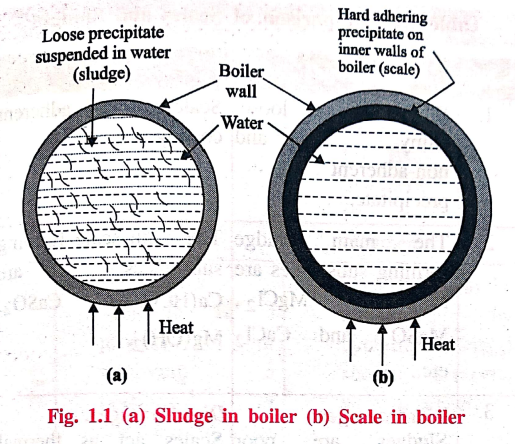
1. Sludge (Loose deposit)
If the precipitate is
loose and slimy it is called sludge. Sludges are formed by the substances like
MgCl2, MgCO3, MgSO4 and CaCl2. They
have greater solubilities in hot water than cold water.
2. Scale (Hard deposit)
On the other hand, if
the precipitate forms hard and adherent coating on the inner walls of the
boiler, it is called scale. Scales are formed by substances like Ca(HCO3)2,
CaSO4 and Mg(OH)2.

Disadvantages of Scale and Sludge Formation
1.
Wastage of fuels
Scales have low thermal
conductivity, so the heat transfer from boiler to inside water is not
efficient. In order to provide steady supply of heat to water, overheating is
done and this causes wastage of fuel. The wastage of fuel depends on the
thickness and nature of the scale, which is shown in the table.

2.
Decrease in efficiency
Scales sometimes
deposit in the valves condensers of the boiler and choke. This results in
decrease efficiency of the boiler.
3.
Boiler explosion
Sometimes due to over
heating the thick scales may crack and causes sudden contact of high heated
boiler material with water. This causes formation of a large amount of steam
and high pressure is developed which may lead to explosion.
Remediation (or) Prevention of scale and sludge formation
1. At the initial
stage, scales and sludges can be removed using scraper, wire, brush, etc.,
2. If scales are
brittle, they can be removed by thermal shocks.
3. By using suitable
chemicals like dil. acids (for CaCO3 scale), EDTA (for CaSO4
scale) with which they form suitable complexes.
4. If the scales and
sludges are loosely adhering, they can be removed by frequent blow down
operation.
5. Using softwater in
boilers scales and sludges can be prevented.
2. Priming and Foaming (carry over)
During the production
of steam in the boiler, due to rapid boiling, some droplets of liquid water are
carried along with steam. Steam containing droplets of liquid water is called
wet steam. These droplets of liquid water carry with them some dissolved salts
and suspended impurities. This phenomenon is called carry over. It occurs due
to priming and foaming.
1. Priming
Priming is the process
of production of wet steam.
Causes (or) Reason
Priming is caused by
(i) High steam
velocity.
(ii) Very high water
level in the boiler.
(iii) Sudden boiling of
water.
(iv) Very poor boiler
design.
Remediation (or) Prevention
Priming can be
controlled by
(i) Controlling the
velocity of steam.
(ii) Keeping the water
level lower.
(iii) Good boiler
design.
(iv) Using treated
water.
2. Foaming
The formation of stable
bubbles above the surface of water is called foaming. These bubbles are carried
over by steam leading to excessive priming.
Causes (or) Reason
Foaming is caused by
the
(i) presence of oil,
and grease,
(ii) presence of finely
divided particles.
Remediation (or) Prevention
Foaming can be
prevented by
(i) adding coagulants
like sodium aluminate, aluminium hydroxide,
(ii) adding anti‒foaming
agents like synthetic polyamides.
3. Caustic Embrittlement (Intercrystalline Cracking)
Caustic embrittlement
means intercrystalline cracking of boiler metal.
Boiler water, usually
contains a small proportion of Na2CO3. In high pressure
boilers this Na2CO3 undergoes decomposition to give NaOH.
Na2CO3
+ H2O → 2NaOH + CO2
This NaOH flows into
the minute hair cracks and crevices, usually present on the boiler material, by
capillary action and dissolves the surrounding area of iron as sodium ferroate.
Fe + 2NaOH → Na2FeO2
+ H2↑
This causes brittlement
of boiler parts, particularly stressed parts like bends, joints, rivets, etc.,
causing even failure of the boiler.
Causes
(or) Reason
Presence of Na2CO3
(or) NaOH in the boiler water.
Remediation
(or) Prevention
Caustic embrittlement
can be prevented by
(i) using sodium
phosphate as softening agent instead of sodium carbonate.
(ii) by adding tannin, lignin
to the boiler water, which blocks the hair cracks.
4. Boiler corrosion
Corrosion in boilers is
due to the presence of
1. dissolved oxygen.
2. dissolved carbon
dioxide.
3. dissolved salts.
1. Dissolved oxygen (DO)
Dissolved oxygen in
water is mainly responsible for the corrosion of boiler. The dissolved oxygen
in water attacks the boiler material at higher temperature.
4Fe + 6H2O +
302 → 4Fe(OH)3 ↓
Remediation
(or) Removal of dissolved oxygen
Dissolved oxygen can be
removed by chemical (or) mechanical methods.
(a)
Chemical method
Sodium sulphite,
hydrazine are some of the chemicals used for removing dissolved oxygen.
2Na2SO3
+ O2 → 2Na2SO4
N2H4
+ O2 → N2+2H20
Hydrazine is found to
be an ideal compound for removing dissolved oxygen in the water, since the
products are water and inert N2 gas.
(b)
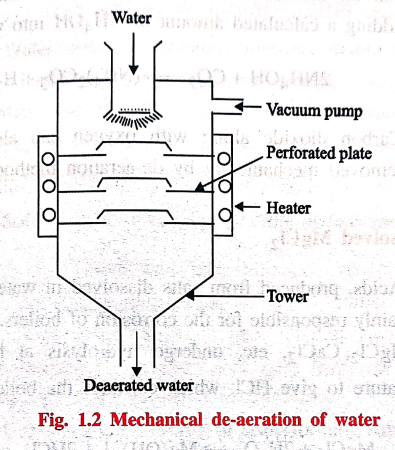
Mechanical de‒aeration
Dissolved oxygen can
also be removed from water by mechanical deaeration (Fig. 1.2).
In this process, water
is allowed to fall slowly on the perforated plates fitted inside the tower. The
sides of the tower are heated, and a vacuum pump is also attached to it. The high
temperature and low pressure produced inside the tower reduce the dissolved
oxygen content of the water.
2. Dissolved carbon dioxide
Dissolved carbon
dioxide in water produces carbonic acid, which is acidic and corrosive in
nature
CO2 + H2O
→ H2CO3
Carbon dioxide gas is
also produced from the decomposition of bicarbonate salts present in water.
Ca(HCO3)2
‒‒Δ→ CaCO3↓ + H2O + CO2↑
Removal
of dissolved Carbon dioxide
(a) Carbon dioxide can
be removed from water by adding a calculated amount of NH4OH into
water.
2NH4OH + CO2
→ (NH4)2CO3 + H2O
(b) Carbon dioxide
along with oxygen can also be removed mechanically by de‒aeration method.
3. Dissolved MgCl2
Acids, produced from
salts dissolved in water, are also mainly responsible for the corrosion of boilers.
Salts like MgCl2, CaCl2, etc, undergo hydrolysis at
higher temperature to give HCl, which corrodes the boiler.
MgCl2 + 2H2O
→ Mg(OH)2↓ + 2HCl
Fe + 2HCl → FeCl2
+ H2↑
FeCl2 + 2H2O
→ Fe(OH)2 + 2HCI
Removal
of acids by neutralisation
Corrosion by acids can
be avoided by the addition of alkali to the boiler water.
HCl + NaOH → NaCl + H2O
Applied Chemistry I: UNIT 1: Water Technology : Tag: Applied Chemistry : - Troubles of Industrial Feed Water Containing Impurities (hardness) (or) Boiler Troubles
Applied Chemistry I: UNIT 1: Water Technology
Under Subject
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Related Subjects
English Essentials I
EN25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
தமிழர் மரபு - Heritage of Tamils
UC25H01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Calculus
MA25C01 Maths 1 M1 - 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I
PH25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Makerspace
ME25C04 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming C
CS25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming Python
CS25C02 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Fundamentals of Electrical and Electronics Engineering
EE25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Mechanical Engineering
ME25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Civil Engineering
CE25C01 1st Semester Civil Department | 2025 Regulation | 1st Semester 2025 Regulation
Essentials of Computing
CS25C03 1st Semester - AID CSE IT Department | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I Laboratory
PH25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Applied Chemistry I Laboratory
CY25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming C Laboratory
CS25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming Python Laboratory
CS25C02 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Engineering Drawing
ME25C01 EEE Mech Dept | 2025 Regulation | 2nd Semester 2025 Regulation
Basic Electronics and Electrical Engineering
EE25C04 1st Semester ECE Dept | 2025 Regulation | 2nd Semester 2025 Regulation

















