Applied Chemistry I: UNIT 4: Corrosion and Control
Corrosion and Control: Important Two Marks Questions and Answers
Important part-A 2 marks Short Questions and Answers - Applied Chemistry I: UNIT 4: Corrosion and Control
UNIT 4: Corrosion and
Control
Two Marks Questions and Answers
1.
What is corrosion? What are its types.
Corrosion is defined as
the gradual destruction or deterioration of metals or alloys by the chemical or
electrochemical reaction with its environment.
Based on the
environment, corrosion is classified into
(i) Dry (or) chemical
corrosion, and
(ii) Wet (or)
electrochemical corrosion.
2.
What is dry corrosion? Give one example.
Dry corrosion is the
corrosion occurs due to the attack of metal surfaces by the atmospheric gases
such as oxygen, hydrogen sulphide, sulphur dioxide, nitrogen, etc.
Example: Oxidation
corrosion
Anode: M → M2+
+ 2e‒
Cathode: ½ O2
+ 2e− → O2‒
Overall reaction: M + ½O2
→ M2+ + O2‒
3.
Explain the consequences of corrosion.
1. Due to formation of
corrosion product over the machinery, the efficiency of the machine gets lost.
2. The products gets
contaminated due to corrosion.
3. The corroded
equipment must be replaced frequently.
4. Plant gets failure
due to corrosion.
5. It is necessary for
over design to compensate for the corrosion.
6. Corrosion releases
toxic products, health hazard, etc.
4.
State Pilling‒Bedworth rule. What is its importance.
(a) According to
Pilling‒Bedworth rule, if the volume of the oxide layer formed is less than the
volume of the metal, the oxide layer is porous and non‒protective.
(b) On the other hand,
if the volume of the oxide layer formed is greater than the volume of the
metal, the oxide layer is non‒porous and protective.
Importance:
Protective and non‒protective
nature of the oxide film can be found out.
5.
How does the liquid metal cause corrosion on metals?
(i) Either dissolution
of a solid metal by a liquid metal. (or)
(ii) Liquid metal may
penetrate into the solid metal.
6.
What is meant by hydrogen embrittlement?
Formation of cracks and
blisters by the hydrogen gases, when it contacts to H2S, is called
hydrogen embrittlement. Hydrogen embrittlement causes loss in ductility of a
material.
7.
What is decarburisation?
The process of decrease
in carbon content in steel is termed as "decarburisation" of steel.
8.
What is electrochemcial corrosion.
Electrochemical
corrosion is the one which occur,
(i) When two dissimilar
metals or alloys are in contact with each other in presence of an aqueous
solution or moisture.
(ii) When a metal is
exposed to varying concentration of oxygen or any electrolyte.
9.
In electrochemical corrosions what is the general anodic reaction of a metal M?
What is the cathodic reaction in basic aqueous solution containing dissolved
oxygen?
Anodic reaction: M → M2+
+ 2e ̄
Cathodic reaction: ½O2
+ H2O + 2e ̄ → 2OH‒
10.
What is galvanic cell corrosion.
(or)
Explain
galvanic corrosion with suitable examples.
When two different
metals are in contact with each other in presence of an aqueous solution (or)
moisture, galvanic corrosion occurs.
Here the more active
metal acts as anode and the less active metal acts as cathode.
Example:
In Zn‒Fe couple, Zn
acts as anode and undergoes corrosion.
But in Fe‒Cu couple, Fe
acts as anode and undergoes corrosion.
11.
How is galvanic corrosion prevented?
Galvanic corrosion is
prevented by
(a) selecting the metals
as close as possible in the electrochemical series.
(b) providing smaller
area for cathode and larger area for anode.
(c) inserting an insulating
material between the two metals.
12.
Steel screw in a brass marine hardware corrodes ‒ Explain.
This is due to galvanic
corrosion. Iron (higher position in electrochemical series) becomes anodic and
is attacked and corroded, while brass (lower in electrochemical series) acts as
cathodic and is not attacked.
13.
Bolt and nut made of the same metal is preferred in practice. Why?
It is preferred in
practice, because galvanic corrosion is avoided due to homogeneous metals (no
sigma anodic and cathodic part).
14.
What is differential aeration corrosion
Differential aeration
corrosion is a concentration cell corrosion, occurs when a metal is exposed to
varying concentration of oxygen (or) air.
15.
Corrosion of a metal is the highest at the metal junction in a galvanic couple.
Account the reason.
Metal junction, where
air cannot diffuse easily, is less aerated and becomes anodic, the remaining
parts are more aerated and becomes cathodic. So, the corrosion concentrates at
the metal junction in a galvanic couple.
16.
What is meant by differential metal corrosion? Explain with an example.
Differential metal
corrosion is nothing but galvanic corrosion, which occurs when two different
metals are joint (or) if the pure metal contains impurity.
The corrosion on metal
depends on their relative position in the emf series. If the impurity (one
metal) is less noble than the bulk metal (second metal), the impurity will act
as anode and thus corrodes and vice versa.
17.
Comment the type of corrosion occurring on lead pipeline passing through clay
to cinders.
Lead pipeline passing
through clay to cinders undergo corrosion. Since the pipeline under cinders is
more aerated, it gets corroded easily.
18.
What is pitting corrosion?
Pitting is a localised
attack, resulting in the formation of a hole around which the metal is
relatively unattacked.
Example: Metal area
covered by a drop of water, sand, dust.
19.
How does a drop of water, oil, dust, resting on an iron surface lead to
corrosion of the metal.
The area covered by the
drop of water acts as an anode due to less oxygen concentration and suffers
corrosion. The uncovered area (freely exposed to air) acts as a cathode due to
high oxygen concentration.
20.
Why does corrosion of water on filled steel tanks occur below water line?
What
type of corrosion occurs in the following cases (i) Wire fence (ii) Riveted joints
(iii) Iron tanks, used for storing water.
Differential aeration
or concentration cell type corrosion.
21.
Irons is corroded faster than aluminium even though Fe is placed below Al in electrochemical series. Why?
Al forms a thin, non‒porous, tightly adhering protective film of Al2O3
on its surface and this film does not allow Al to undergo further corrosion.
22.
What is water line corrosion? Explain with an example.
(or)
What
is concentration cell corrosion? Give an example.
Justify
the formation of water line corrosion.
This type of corrosion
occurs when a metal is exposed to varying concentration of oxygen or any
electrolyte or water on the surface of the base metal. Here the metal above the
water is more aerated and become cathodic, the part below the water is less
aerated and become anodic.
Example: Metals
partially immersed in a water (or) conducting solution.
23.
What is differential aeration?
Differential aeration
is nothing but two different concentration of air on a metal surface.
24.
List out the the differences between chemical corrosion and electrochemical
corrosion.

Dry or Chemical
corrosion
1. It occurs in dry
state.
2. It follows
adsorption mechanism.
3. Corrosion product
accumulate on the same spot, where corrosion occurs.
Wet or Electrochemical
corrosion
1. It occurs in
presence of moisture or electrolyte.
2. It follows the
mechanism of electro chemical reaction.
3. Corrosion occurs at anode
while products gather at cathode.
25.
What is deactivation? Give examples.
Deactivation is a process
of removing dissolved oxygen by adding some chemicals in aqueous solution.
Examples
1. Sodium sulphite: 2Na2SO3
+ O2 → 2Na2SO4
2. Hydrazine: N2H4
+ O2 → N2 + 2H2O
26.
Relate the rate of corrosion with temperature.
Rate of corrosion ∝ Temperature.
27.
List the environmental factors that influence corrosion.
1. Temperature
2. Humidity
3. Presence of
corrosive gases
4. Presence of
suspended particles
5. Effect of pH
28.
What are factors which affects corrosion?
1. Position of the
metal in emf series
2. Relative areas of
the anode and cathode
3. Purity of the metal
4. Over voltage
5. Nature of the
surface film
6. Nature of the
corrosion product
7. Temperature &
Humidity
29.
State Pilling Bedworth rule. Name two metals in which the specific volumes of
their oxides are greater than that of the metals.
(a) According to
Pilling‒Bedworth rule, if the volume of the oxide layer formed is less than the
volume of metal, the oxide layer is porous and non‒protective.
(b) On the other hand,
if the volume of the oxide layer formed is greater than the volume of metal,
the oxide layer is non‒porous and protective.
Ex: Volume of Oxides of
Pb & Sn are greater than that of metals.
30.
Using chemical equations, state the mechanism of corrosion of iron in weakly
alkaline solution.
When an iron metal
contacts with a neutral (or) weakly alkaline solution of an electrolyte in
presence of oxygen, OH‒ ions are formed.
At anode
Iron dissolves as Fe2+
with the liberation of electrons.
Fe → Fe2+ +
2e‒ (oxidation)
At cathode
The liberated electrons
flow from anodic to cathodic part through metal, where the electrons are taken
up by the dissolved oxygen to form OH‒ ions.
½ O2 + H2O + 2e‒
→ 2OH‒
Thus, the net corrosion
reaction is
Fe2+ + 2OH‒ → Fe(OH)2↓
If enough O2
is present Fe(OH)2 is easily oxidized to Fe(OH)3, a rust
(Fe2O3. H2O).
4Fe(OH)2 + O2
+ 2H2O → 4Fe(OH)3
31.
Explain why magnesium corrodes faster when it is in contact with copper than
when it is in contact with iron.
The Magnesium corrodes
faster when it is in contact with copper because larger difference in their
standard reduction potential values between Mg and Cu. Mg have (‒) ve reduction
potential value and Cu have (+) ve reduction potential value.
(Mg = ‒2.3 V; Cu = + 0.34 V)
But, lesser difference
in the standard reduction potential values between Mg and Fe and both have (‒)
ve reduction potential values.
(Mg = ‒ 2.37 V; Fe ‒ 0.44 V)
32.
Why does Mg corrode faster than iron?
Mg possess higher (‒)ve
reduction potential (E° = ‒ 2.37 V) than the iron (E° = ‒0.44 V). So Mg corrodes
faster.
33.
Zinc is more readily corroded when coupled with copper than with lead why?

Zinc is anodic to Cu
and Pb. When zinc is (Eo = ‒0.76 V) is coupled with copper (E° =
+0.34 V) zinc is more readily corroded, because the difference in their
possition in emf series is more. Also Cu has (+)ve reducetion potential. But,
if Zn is coupled, with lead (E° = ‒0.13 V) rate corrosion is less because the
difference in their possition in emf series is less. Also Pb has (‒)ve
reduction potential.
34.
Zinc reacts with dilute sulphuric acid give hydrogen but silver doesn't
liberate hydrogen. Why?
Metals with negative
reduction potential (like Zn) (i.e., the metals placed above H2 in
the emf series) liberates the hydrogen from an acid solution.
Zn + H2SO4 → ZnSO4
+ H2↑
E°Zn = ‒0.76 volt
But, the silver with
positive reduction potential (i.e., the metals placed below H2 in
the emf series) does not liberate the hydrogen from an acid solution.
Ag + H2SO4
→ No reaction
E°Ag = +0.80
volt
35.
Small anodic area results in intense corrosion. Why?
Rate of corrosion is
more rapid and intense, if the anodic area is smaller and the cathodic area is
larger. When the cathodic area is larger, the demand for electrons will be more
and this results in an increased rate of corrosion of metals at anodic area.
36.
Why corrosion rate increases with temperature?
The rate of chemical
reaction and rate of diffusion of the ions increases with rise of temperature.
37.
What is a sacrificial anode? How does it protect a submerged pipeline?
A sacrificial anode is
more active metal than the anodic part of the corrosion cell.
The sacrificial anode
is connected to the submerged pipeline. So the submerged pipeline will be
converted to cathode and corrosion concentrates over the sacrificial anode.
38.
Which of the following metals could provide cathodic protection to iron: Al,
Zn, Cu, Ni
Al and Zn (Both are
placed above the Fe in emf series)
39.
How does impressed current cathodic method carried out?
(or)
What
is the principle involved in impressed cathodic current method of prevention of
corrosion?
In impressed current
cathodic protection, current is applied in the opposite direction of the
corrosion current to nullify it.
40.
Compare sacrificial anode method and impressed current method.
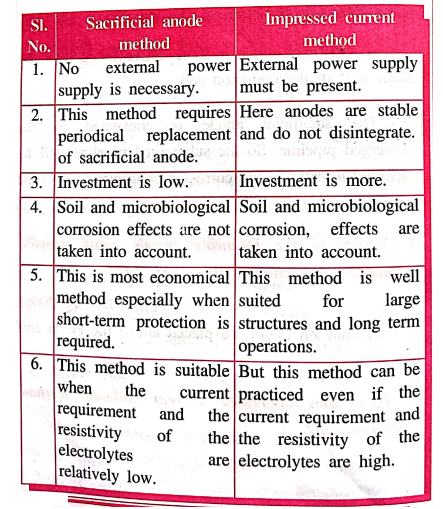
Sacrificial anode
method
1. No external power
supply is necessary.
2. This method requires
periodical replacement of sacrificial anode.
3. Investment is low.
4. Soil and
microbiological corrosion effects are not taken into account.
5. This is most
economical method especially when short‒term protection is operations.
6. This method is
suitable when the current requirement and the resistivity of the electrolytes are
relatively low.
Impressed current
method
1. External power
supply must be present.
2. Here anodes are
stable and do not disintegrate.
3. Investment is more.
4. Soil and
microbiological corrosion, effects are taken into account.
5. This method is well suited
for large structures and long term operations.
6. But this method can
be practiced even if the current requirement and the resistivity of the electrolytes
are high.
41.
Write the effetct of pH of the conducting medium on corrosion of metals.
When pH of the
conducting medium increases, rate of corrosion increases.
pH ∝
corrosion rate
42.
Give the importance of pilling‒bedworth rule.
1. Rate of dry
corrosion can be predicted.
2. Protective or non‒protective
nature of the oxide film can be found out.
43.
Define glavanic series.
A galvanic series is a
chart (or) list that orders metals and alloys based on their oxidation
potential (activity) in a particular environment (seawater (or) soil)
44.
Mention any two applications of galvanic series.
1. It is used to
prevent galvanic corrosion.
2. Engineers use the
series to select materials that are close to each other to minimize corrosion.
3. It is used to
prevent corrosion of ship hulls and other metallic components.
45
How is ceramic coating produced?
Ceramic coatings are
made of mixing high refractory oxides such as chromium trioxide with silica materials.
The paste of silica material and refractory oxide is sprayed and then fired.
46.
What are enamels?
Enamels are pigmented
varnish, ie., a colloidal dispersion of pigments and varnish. Enamels dry
slowly but form hard, lustrous and glossy film.
47.
What are the uses of enamels?
1. Enamels are used to
protect the materials against corrosion.
2. Japans are used for
painting bicycles, automobiles, etc.,
48.
What are the requisites of a good paint?
(i) It should spread
easily on the metal surface.
(ii) It should have
high hiding (covering) power.
(iii) It should not
crack on drying.
(iv) It should adhere
well to the surface.
(v) The colour of the
paint should be stable.
(vi) It should be a
corrosion and water resistant.
(vii) It should give a
glossy film.
49.
What is the role of pigment in paint? Give two examples.
Pigments are solid and
colour producing substances in the paint.
White pigments ‒ White
lead, lithophone.
Black pigments ‒ Lamp
black, carbon black.
50.
Explain the functions of driers in the paint.
(i) They act as oxygen
carriers (or) catalysts.
(ii) They provide
oxygen, which is essential for oxidation, polymerisation of drying oil.
Examples: Metallic
soaps, linoleats and resinates of Co, Mn and Pb.
51.
What is the function of extender in the paint?
(i) It reduces the cost
of the paint.
(ii) It retards the
setling of the pigment in all paints.
(iii) It modifies the
shades of the pigments.
(iv) It prevents
shrinkage and cracking.
Examples: Talc, gypsum,
chinaclay, etc.
52.
What is mechanism of drying of oil paint?
The mechanism involves
oxidation, polymerisation and condensation reactions. The oil containing
conjugated double bonds dry very faster than the non‒conjugated oils.
53.
What are the structural arrangement required for drying of a drying oil?
Conjugated oil dry very
faster than non‒conjugated oil.
(i) Conjugated oil
structure
‒CH=CH‒CH=CH‒CH=CH‒
(ii) Non‒conjugated oil
structure:
‒CH=CH‒CH2‒CH=CH
‒
54.
How to calculate P.V.C of paint? Mention its significance.
It is an important
property of a paint. The following equation is used to calculate the P.V.C.
P.V.C = Volume of
pigment in the paint / [ Volume of pigment in the paint + Volume of non‒volatile
vehicle in the paint ]
Higher the volume of
P.V.C, lower will be the durability, adhesion, consistency of the paint.
55.
What are the reasons for a failure of a paint?
1. Chalking
2. Cracking
3. Erosion
4. Blistering.
56.
What is a varnish?
Varnish is a
homogeneous colloidal solution of natural or synthetic resins in spirit or oil.
Varnish is used for both protection and decoration of metal surfaces.
57.
How is an oil varnish prepared?
Oil varnish is prepared
by dissolving natural or synthetic resins in a drying oil and volatile solvent.
This type of varnish dries by the evaporation of the solvent followed by
oxidation and polymerisation of the drying oil.
58.
Explain the characteristics of a good varnish.
(i) It should be soft.
(ii) It should produce
a shining and glossy film on drying.
(iii) It should dry
quickly.
(iv) It should not
shrink or crack after drying.
59.
What are the applications of varnishes?
1. It is used to
protect the materials against corrosion.
2. It is a coating
prier to paint coating.
3. It is also used for
improving the appearance of wooden surfaces.
60.
How do paints differ from varnishes?
(i) Paint: Contains
pigments- Vamish: Does not contain pigments.
(ii) Paint: Opaque,
colloidal solution - Vamish: Transparent, homogenous solution.
61.
What is a lacquer? How does it differ from varnish. Mention its uses.
Lacquer is a colloidal
dispersion of cellulose derivatives, resins and plasticiers in solvents and
diluents. It is used for interior decoration. It does not contain drying oil
like varnish.
62.
What are Japans?
When coloured pigments
are used, such enamels are called japans. If black pigments are used, the
enamels are called black japans.
63.
What is anodising?
Anodising (or) anodic
oxidation is an electrolytic process, in which a thick oxide coating is
produced on the base metal.
64.
What are the advantages of anodization?
(i) an insulating coat
for the electrically conducting base metal (aluminium).
(ii) very good
resistance to corrosion,
(iii) thicker oxide
coating and hence it is more protective,
(iv) the film may be
coloured with organic (or) inorganic dyes,
(v) anodized metal
(like anodised aluminium) is used in automobile engine piston.
65.
How is anodising of aluminium carried out?
Anodised coating on
aluminium is done by making aluminium as an anode in an electrolytic bath of H2SO4
(or) oxalic acid and the cathode is a plate of lead or stainless steel.
66.
Why is anodising is best suited to aluminium?
Since aluminium has
high negative reduction potential it can undergo oxidation readily in presence
of electrolyte like chromic acid. By making aluminium as anode in the
electrolytic cell, a thick oxide coating can be produced on its surface.
67.
What is the difference between chromising and chromate coating?
Chromising is
cementation, obtained by heating the base metal with chromium powder.
Chromate coating is
produced by dipping the metal object in a bath of acidic potassium chromate,
followed by dipping in neutral chromate solution.
Applied Chemistry I: UNIT 4: Corrosion and Control : Tag: Applied Chemistry : - Corrosion and Control: Important Two Marks Questions and Answers
Applied Chemistry I: UNIT 4: Corrosion and Control
Under Subject
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Related Subjects
English Essentials I
EN25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
தமிழர் மரபு - Heritage of Tamils
UC25H01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Calculus
MA25C01 Maths 1 M1 - 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I
PH25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Makerspace
ME25C04 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming C
CS25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming Python
CS25C02 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Fundamentals of Electrical and Electronics Engineering
EE25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Mechanical Engineering
ME25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Civil Engineering
CE25C01 1st Semester Civil Department | 2025 Regulation | 1st Semester 2025 Regulation
Essentials of Computing
CS25C03 1st Semester - AID CSE IT Department | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I Laboratory
PH25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Applied Chemistry I Laboratory
CY25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming C Laboratory
CS25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming Python Laboratory
CS25C02 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Engineering Drawing
ME25C01 EEE Mech Dept | 2025 Regulation | 2nd Semester 2025 Regulation
Basic Electronics and Electrical Engineering
EE25C04 1st Semester ECE Dept | 2025 Regulation | 2nd Semester 2025 Regulation

















