Applied Chemistry I: UNIT 4: Corrosion and Control
Factors Influencing the rate of corrosion
The rate and extent of corrosion mainly depends on the following two factors. 1. Nature of the metal 2. Nature of the environment
FACTORS INFLUENCING THE RATE OF
CORROSION
The rate and extent of
corrosion mainly depends on the following two factors.
1. Nature of the metal
2. Nature of the
environment
1. Nature of the metal
1. Position in emf series
Extent of corrosion
depends on the position of the metal in the emf series. Metals above the
hydrogen in emf series get corroded vigorously. Lower the reduction potential,
greater is the rate of corrosion. When two metals are in electrical contact,
the more active metal (or the metal having high negative reduction potential)
undergoes corrosion.
The rate and severity
of corrosion depends on the difference in their positions in the emf series.
Greater the difference faster is the corrosion rate.
2. Relative areas of the anode and cathode
Rate of corrosion will be more, when the cathodic area is larger. When the cathodic area is larger, the demand for electrons will be more and this results in an increased rate of corrosion (dissolution) of metals at anodic area.
3. Purity of the metal
100% pure metal will
not undergo any type of corrosion. But, the presence of impurities in a metal
create heterogeneity and thus galvanic cells are set up with distinct anodic
and cathodic area in the metal. Higher the percentage of impurity, faster is
the rate of corrosion of the anodic metal.
The effect of impurities
on the rate of corrosion of zinc is given below.

4. Over voltage or over potential
Over voltage of a metal
in the corrosive environment is inversely proportional to corrosion rate.
Example: The normal
hydrogen over voltage of zinc metal, when it is dipped in 1 M H2SO4,
is 0.7 volt. Here the rate of corrosion is low. By adding small amount of
impurity like CuSO4 to H2SO4, the hydrogen
over voltage is reduced to 0.33 V. This results in increased rate of corrosion
of zinc metal.
5. Nature of the surface film
Nature of the oxide
film, formed on the metal surface, decides the extent of corrosion which can be
decided by Pilling‒Bedworth rule
(i) In case of alkali
and alkaline earth metals such as Mg, Ca, etc. form oxide, whose volume is
lesser than the volume of metal. Hence the oxide film will be porous and non‒protective
and bring about further corrosion.
(ii) But in heavy
metals like Al, Cr, etc. form oxide, whose volume is greater than that of the
metal. Hence the oxide film will be non‒porous and protective and prevents
further corrosion.
6. Nature of the corrosion product
If the corrosion
product is soluble in the corroding medium, the corrosion rate will be faster.
Similarly, if the corrosion product is volatile (like MoO3 on Mo
surface), the corrosion rate will be faster.
2. Nature of the environment
1. Temperature
Rate of corrosion is
directly proportional to temperature. This is because, the rate of chemical
reaction and the rate of diffusion of the ions increases with rise in
temperature. Hence the rate of corrosion increases with temperature.
2. Humidity
Rate of corrosion will
be more, when the humidity in the environment is high. The moisture acts as a
solvent for the oxygen in the air to produce the electrolyte, which is essential
for setting up a corrosion cell.
3. Presence of corrosive gases
Acidic gases like, CO2,
SO2, H2S and fumes of HCI, H2SO4,
etc., produce electrolytes, which are acidic and increases the electrochemical
corrosion.
4. Presence of suspended particles
Particles like, NaCl,
(NH4)2SO4 along with moisture act as powerful
electrolytes and thus accelerate the electrochemical corrosion.
5. Effect of pH
Possibility of
corrosion with respect to pH of the electrolytic solution and the electrode
potential of the metal is correlated with the help of a pourbaix diagram.
Pourbaix
diagram
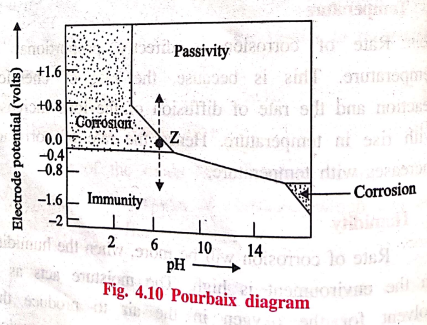
The pourbaix diagram
for iron in water is shown in fig. 4.10. It shows clearly the zones of
corrosion, immunity and passivity. In the diagram, 'Z' is the point where pH =
7 and the electrode potential = ‒0.4 V. It is present in the corrosion zone.
This clearly shows that iron rusts in water under those conditions. In actual
practice, it is observed to be true.
From the diagram (Fig.
4.10) it is clear that the rate of corrosion can be altered by shifting the
point 'Z' into immunity or passivity regions. The iron will be immune to
corrosion, if the potential is changed to about ‒0.8 V by applying external
current. On the other hand, the rate of corrosion of iron can also be reduced
by moving into the passivity region by applying positive potential.
The diagram clearly
indicates that the rate of corrosion can also be reduced by increasing the pH
of the solution by adding alkali.
Thus, the rate of
corrosion will be maximum when the corrosive environment is acidic. i.e., pH is
less than 7.
Applied Chemistry I: UNIT 4: Corrosion and Control : Tag: Applied Chemistry : - Factors Influencing the rate of corrosion
Applied Chemistry I: UNIT 4: Corrosion and Control
Under Subject
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Related Subjects
English Essentials I
EN25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
தமிழர் மரபு - Heritage of Tamils
UC25H01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Calculus
MA25C01 Maths 1 M1 - 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I
PH25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Makerspace
ME25C04 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming C
CS25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming Python
CS25C02 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Fundamentals of Electrical and Electronics Engineering
EE25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Mechanical Engineering
ME25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Civil Engineering
CE25C01 1st Semester Civil Department | 2025 Regulation | 1st Semester 2025 Regulation
Essentials of Computing
CS25C03 1st Semester - AID CSE IT Department | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I Laboratory
PH25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Applied Chemistry I Laboratory
CY25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming C Laboratory
CS25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming Python Laboratory
CS25C02 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Engineering Drawing
ME25C01 EEE Mech Dept | 2025 Regulation | 2nd Semester 2025 Regulation
Basic Electronics and Electrical Engineering
EE25C04 1st Semester ECE Dept | 2025 Regulation | 2nd Semester 2025 Regulation

















