Applied Chemistry I: UNIT 4: Corrosion and Control
Wet (or) Electro-chemical Corrosion
Mechanism , Types
Mechanism of wet corrosion, Types of electrochemical corrosion
WET OR ELECTRO-CHEMICAL CORROSION
Wet corrosion occurs
under the following conditions.
(i) When two dissimilar
metals or alloys are in contact with each other in the presence of an aqueous solution
or moisture.
(ii) When a metal is
exposed to varying concentration of oxygen or any electrolyte.
Mechanism of wet corrosion
Under the above
conditions, one part of the metal becomes anode and another part becomes
cathode.
1.
At anode
In anodic part,
oxidation (or) dissolution of metal occurs
M → M2+ + 2e‒
2.
At cathode
In cathodic part
reduction reaction occurs, which depends on nature of the corrosive
environment.
(a)
Acidic environment
If the corrosive
environment is acidic, hydrogen evolution occurs at cathodic part.
2H+ + 2e‒→
H2↑
(b)
Neutral environment
If the corrosive
environment is slightly alkaline (or) neutral, hydroxide ions are formed at
cathodic part.
½ O2 + 2e‒
+ H2O → 2OH‒
Thus, the metal ions
(from anodic part) and non‒metallic ions (from cathodic part) diffuse towards
each other through conducting medium and form a Corrosion product between anode
and cathode.
(a) Hydrogen evolution type corrosion
"All metals above
hydrogen in the electrochemical series have a tendency to get dissolved in
acidic solution with simultaneous evolution of hydrogen gas" (Fig. 4.2).
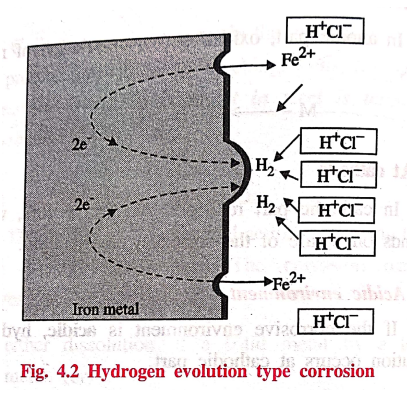
Example: When iron
metal contacts with non‒oxidising acid like HCl, H2 evolution
occurs.
At
anode
Iron undergoes
dissolution to give Fe2+ ions with the liberation of electrons.
Fe → Fe2+ +
2e‒ (oxidation)
At
cathode
The liberated electrons
flow from anodic to cathodic part, where H+ ions get reduced to H2.
2H+ + 2e ̄ → H2↑ (reduction)
(b) Absorption of oxygen (or) Formation of hydroxide ion type corrosion
The surface of iron is
usually, coated with a thin film of iron oxide. However, if the oxide film
grows, some crack will form and anodic areas are created on the surface while
the remaining part acts as cathode (Fig. 4.3).
Example:
When iron metal
contacts with a neutral (or) slightly alkaline solution of an electrolyte in
presence of oxygen, OH‒ ions are formed.

At
anode
Iron, dissolves as Fe2+
with the liberation of electrons.
Fe → Fe2+
+ 2e‒ (oxidation)
At
cathode
The liberated electrons
flow from anodic to cathodic part through metal, where the electrons are taken
up by the dissolved oxygen to form OH‒ ions.
½O2 + H2O + 2e‒
→ 2OH‒
Thus, the net corrosion
reaction is
Fe2+ + 2OH ̄ → Fe(OH)2↓
If enough O2
is present, Fe(OH)2 is easily oxidized to Fe(OH)3, a rust
(Fe2O3. H2O).
4Fe(OH)2 + O2
+ 2H2O → 4Fe(OH)3 (or)
(2Fe2O3.3H2O)
brown rust
Table
4.1 Difference between Chemical and Electro chemical Corrosion

Chemical Corrosion
1. It occurs only in
dry condition.
2. It is due to the
direct chemical attack of metal by the environment.
3. Even a homogeneous
metal surface gets corroded.
4. Corrosion products
accumulate in the same place, where corrosion occurs.
5. Chemical corrosion
is self‒controlled.
6. It follows
adsorption mechanism.
7. Examples: Formation
of mild scale on iron surface.
Electrochemical Corrosion
1. It occurs in
presence of moisture or electrolyte.
2. It is due to the set
up of a large number of cathodic and anodic areas.
3. Heterogeneous
surface (or) Bimetallic contact is the condition.
4. Corrosion occurs at
the anode, while products formed elsewhere.
5. Electrochemical corrosion
is continuous process.
6. It follows electrochemical
reaction.
7. Examples: Rusting of
iron in moist atmosphere.
1. Types of electrochemical corrosion
1. Galvanic corrosion
When two different
metals are in contact with each other in presence of an aqueous solution or
moisture, galvanic corrosion occurs.
Here, the more active
metal (with more negative electrode potential) acts as anode and the less
active metal (with less negative electrode potential) acts as cathode.
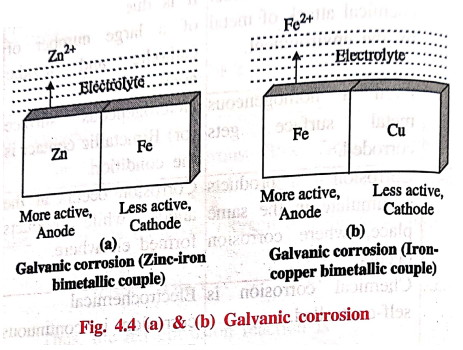
Example: Zn‒Fe couple
Fig. 4.4(a) represents
Zn‒Fe couple, in which zinc (more active or higher in emf series) dissolves in
preference to iron (less active metal) i.e., Zn acts as anode and undergoes
corrosion and Fe acts as cathode.
Example: Cu‒Fe couple
Fig. 4.4 (b) represents
Fe ‒ Cu couple, in which iron (more active, when compared to Cu) dissolves in
preference to copper (less active) i.e., Fe acts as anode and undergoes
corrosion and Cu acts as cathode.
Examples for Galvanic Corrosion
(i)
Steel screw in a brass marine hardware corrodes
This is due to galvanic
corrosion. Iron (higer position in electrochemical series) becomes anodic and
is attacked and corroded, while brass (lower in electrochemical series) acts as
cathodic and is not attacked.
(ii)
Bold and Nut made of the same metal is preferred
It is preferred in
practice, because galvanic corrosion is avoided due to homogeneous metals (no anodic
and cathodic part).
Prevention
Galvanic corrosion can
be minimised by the following two ways.
1. By providing an
insulating material between the two metals.
2. By selecting two
metals as close as possible on the emf series.
2. Differential aeration (or) concentration cell corrosion
This type of corrosion
occurs when a metal is exposed to varying concentration of oxygen or any
electrolyte on the surface of the base metal.
Example: Metals
partially immersed in water (or) conducting solution (called water line
corrosion).
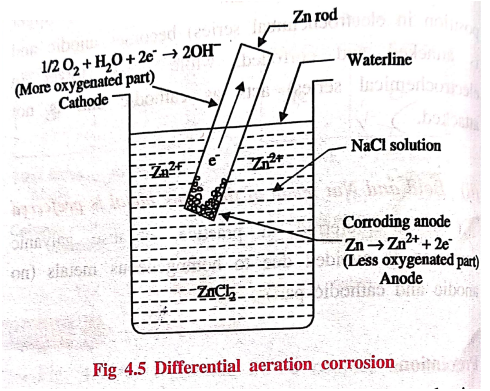
If a metal is partially
immersed in a conducting solution (Fig. 4.5) the metal part above the solution
is more aerated and hence become cathodic. On the other hand, the metal part
inside the solution is less aerated and thus, become anodic and suffers
correcsion.
At
anode (less aerated part): Corrosion occurs
M → M2+ + 2e‒
At
cathode (more aerated part): OH‒ ions
are produced
½O2 + H2O + 2e‒
→ 2OH‒
Examples for Differential aeration corrosion
(a) Pitting or
localised corrosion.
(b) Crevice corrosion.
(c) Pipeline corrosion.
(d) Corrosion on wire
fence.
(a) Pitting corrosion
Pitting is a localised
attack, resulting in the formation of a hole around which the metal is
relatively unattacked.
Example: Metal area
covered by a drop of water, sand, dust, scale, etc.
Let us consider a drop
of water or aqueous NaCl resting on a metal surface (Fig. 4.6). The area
covered by the drop of water acts as an anode due to less oxygen concentration
and suffers corrosion. The uncovered area (freely exposed to air) acts as a
cathode due to high oxygen concentration.
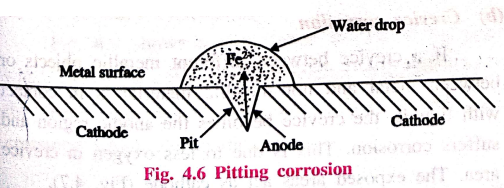
The rate of corrosion will be more, when the area of cathode is larger and the area of anode is smaller, Therefore, more and more material is removed from the same spot. Thus a small hole or pit is formed on the surface of the metal.
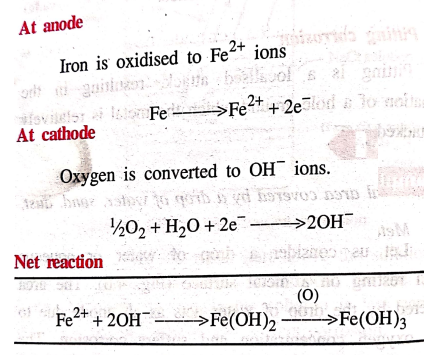
At
anode
Iron is oxidised to Fe2+
ions
Fe → Fe2+ +
2e‒
At
cathode
Oxygen is converted to
OH‒ ions.
½O2 + H2O
+ 2e ̄ → 2OH‒
Net
reaction
Fe2+ + 2OH‒
→ Fe(OH)2 –(O)→ Fe(OH)3
This type of intense
corrosion is called pitting.
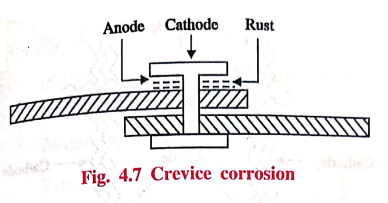
(b) Crevice corrosion
If a crevice between
different metallic objects of between metal and non‒metallic material is in
contact with liquids, the crevice becomes the anodic region and suffers
corrosion. This is due to less oxygen in crevice area. The exposed areas act as
cathode (Fig. 4.7).
(c) Pipeline corrosion
Differential aeration
corrosion may also occur in different parts of pipeline.
Buried pipelines or
cables passing from one type of soil to another say, from clay (less aerated)
to sand (more aerated) may get corroded due to differential aeration.

(d) Corrosion on wire‒fence
Fig. 4.9 shows a wire
fence in which the areas where the wires cross are less aerated than the rest
of the fence and hence corrosion occurs at the wire Crossings, which are
anodic.

Other examples for differential aeration corrosion
1. Corrosion occurring
under metal washers, where oxygen cannot diffuse easily.
2. Lead pipeline
passing through clay to cinders undergo corrosion. Since the pipeline under
cinders is more aerated, it gets corroded easily.
Applied Chemistry I: UNIT 4: Corrosion and Control : Tag: Applied Chemistry : Mechanism , Types - Wet (or) Electro-chemical Corrosion
Applied Chemistry I: UNIT 4: Corrosion and Control
Under Subject
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Related Subjects
English Essentials I
EN25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
தமிழர் மரபு - Heritage of Tamils
UC25H01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Calculus
MA25C01 Maths 1 M1 - 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I
PH25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Makerspace
ME25C04 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming C
CS25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming Python
CS25C02 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Fundamentals of Electrical and Electronics Engineering
EE25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Mechanical Engineering
ME25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Civil Engineering
CE25C01 1st Semester Civil Department | 2025 Regulation | 1st Semester 2025 Regulation
Essentials of Computing
CS25C03 1st Semester - AID CSE IT Department | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I Laboratory
PH25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Applied Chemistry I Laboratory
CY25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming C Laboratory
CS25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming Python Laboratory
CS25C02 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Engineering Drawing
ME25C01 EEE Mech Dept | 2025 Regulation | 2nd Semester 2025 Regulation
Basic Electronics and Electrical Engineering
EE25C04 1st Semester ECE Dept | 2025 Regulation | 2nd Semester 2025 Regulation

















