Applied Physics I: Chapter 10: Applied Optics - Lasers
Lasers: Important Solved Problems
Anna University Solved Problems, Additional Solved Problems, Assignment Problems - Questions with solved Solution and Answer - Applied Physics I: Chapter 10: Applied Optics - Lasers
ANNA UNIVERSITY SOLVED PROBLEMS
1. A cadmium sulphide
photo detector crystal is irradiated over a receiving area of 4×10‒6
m2 by light of wavelength 0.4×10‒6 m and intensity 200
Watt/m2. Assuming that each quantum generates an electron hole pair,
calculate the number of pairs generated per second.
Solution
Intensity
per unit area = Number of photons × Energy of a photon
Number
of photons = (Intensity × area) / Energy of photon (hv)
=
200×4×10‒6 / {6.625×10‒34×[ 3×108 / 0.4×10−6]}
=
8×10‒4 / 4.96875×10‒19
Number of photons = 1.6100 × 1015
Since
each photon generates an electron‒hole pair, the number of photons is equal to
the number of electron‒hole pairs.
Number of electron hole pairs = 1.6100×1015
2. Calculate the
Wavelength of radiation emitted by an LED made up of a semiconducting material
with band gap energy 2.8 eV.
Solution
Given
Eg
= 2.8 eV
=
2.8 × 1.602 × 10‒19 J
=
4.4856 × 10‒19 J
We
know Eg = hv Joules
Eg
= hc / λ
∴ λ = hc /
Eg
=
6.625×10‒34×3×108 / 4.4856×10‒19
λ = 4430.8 Å (Blue colour)
3. For InP laser diode,
the wavelength of light emission is 1.55 μm. What is its band gap in eV?
Solution
Given
λ = 1.55×10‒6 m,
Eg
= ?
Energy
band gap Eg = hv Joules
Eg
= hc / λ
Eg
= 6.625×10‒34×3×108 / 1.55×10‒6
Eg
= 1.28225×10‒19 Joules
Eg
= 1.28255×10−19 / 1.6×10‒19 eV
=
0.8014 eV
Energy band gap Eg = 0.8014 eV
4. Calculate the number
of photons from green light of mercury (λ = 4961Å) requires to do one joule of
work.
Solution
Given
λ=4961×10‒10m
We
know E = hv =
hc / λ
= 6.625×10‒34×3×108 / 4961×10‒10 Joules
E
= 4.006×10‒19 Joules
Number
of photons required to do one Joule of work (N) = 1 Joules / 4.006×10‒19
Joules
N = 2.4961×1018/m3
5. Calculate the long
wavelength limit of a extrinsic semiconductor if the ionisation energy is 0.02
eV.
Solution
The
ionization energy = 0.02 eV
We
know E = hv= hc / λ
E
= hc / λ
λlong
= hc / E
λlong
= 6.625×10‒34×3×108 / 0.02×1.6×10‒19
λlong = 6.210 × 10‒5
m
6. Calculate the
wavelength of emission from GaAs semiconductor laser whose band gap energy is
1.44 eV (Planck's constant is 6.625×10‒34 Js and charge of an
electron is 1.6×10‒19 C.
Given
data
Band
gap energy Eg = 1.44 eV
(or) 1.44 × 1.6 × 10‒19 Joules
Solution
We
know Band gap energy (Eg) = hv
= hc / λ
(or)
we can write λ = hc / Eg
λ
= 6.625×10‒34×3×108 / 1.44×1.6×10‒19
λ = 8.6263 × 10‒7
m
∴ Wavelength of GaAs laser = 8626.3 Å
7. For a semiconductor
laser, the band gap is 0.8 e V. What is the wavelength of light emitted from
it.
Given
Data:
Eg = 0.8 eV = 0.8 × 1.6×10‒19
Joules
Formula:
Wavelength
λ = hc / Eg
λ
= 6.625×10‒34×3×108 / 0.8×1.6×10‒19
∴ λ=1.5527×10‒6m
The wavelength of light emitted = 1.5527 μm
8. Calculate how many
photons are emitted in each minute in a He‒Ne laser source, which emits light
at a wavelength of 6328Å. The output power of this source is 3mW.
Solution
The
frequency of the photon emitted by the laser beam = c/λ
= 3×108 / 6328×10‒10
v
= 4.74 × 1014 Hz
We
know that the energy of a photon E = hv
(or) E = 6.625 × 10‒34 × 4.74 × 1014
(or)
E = 3.14×10‒19 J
Energy
emitted by the laser = 3mW = 3×10‒3 W
=
3×10‒3 Js‒1
=
3×10‒3×60 J min‒1
The
number of photons emitted per minute = 3×10‒3×60 / 3.14×10‒19
= 5.732 × 1017 photons/minute
ALITER
Intensity
per unit area = No. of photons × Energy of a photon
No.
of photons emitted per second (N) = Intensity per unit area / Energy of a
photon
(or)
No. of photons emitted per second (N) = Power output / Energy of a photon
N
= 3×10‒3 / 3.14×10‒19
=
9.554 × 1015
No.
of photons emitted per minute (N) = 9.554 × 1015 × 60
=
5.732 × 1017 photons/minute
The He‒Ne laser source emits 5.732 × 1017 photons per
minute.
9. He‒Ne laser emits
light at a wavelength of 632.8 nm and has an output power of laser is 5 mW. How
many photons are emitted per second by this laser when operating?
Solution
Given
Data:
Wavelength
(λ) = 632.8 × 10‒9 m
Power
output = 5 × 10‒3 Watts
Formula:
Energy
of a photon E = hv = hc / λ
E
= hc / λ
=
6.625×10‒34×3×108 / 632.8×10‒9
E
= 1.9875×10‒25 / 632.8×10−9
E = 3.1408 × 10‒19 Joules
∴ numbers of photons
emitted per second
N
= Power output / Energy of a photon
N
= 5×10‒3 / 3.1048×10‒9
N
= 1.5919 × 10‒16 photons/second
Number of phtons emitted per second 1.5919 × 10−16
10. A Nd‒YAG laser
emits light at wavelength of 1.063×10‒6m If the output power is 20 W, then how many photons are emitted in
ten minutes when the laser is in operation?
Given
data
λ
= 1.063 × 10‒6 m
P
= 20 W
Formula:
Number
of photons = ( Power output ) / ( Energy
of a photon [hv] )
N
= P / [hv / λ]
N
= λP / hc
N
= [ 1.063 × 10‒6 × 20 ] / [ 6.625 × 10−34 × 3 × 108
]
N
= 2.126×10‒5 / 1.9875×10‒25
N
= 1.0697×1020 per second
∴ For 10 minutes N =
1.0697 × 1020 × 60 × 10
∴ N = 6.418 × 1022
∴ Number of photons emitted in 10 minutes = 6.418 × 1022
11. Prove that laser
action is not possible in optical frequencies under thermal equilibrium. (OR)
Show that the
stimulated emission is not possible for:
(i) Sodium D line at
300° C and
(ii) At optical
frequencies under thermal equilibrium.
Solution
Given
data λ = 5000 Å; T = 300°K
We
know Rsp/Rst = e
hv / KBT‒1

=
e96.01 ‒ 1
Rsp/Rst
= 4.9953 × 1041 ‒ 1
Rsp/Rst ≈ 4.9953 × 1041
Since
the spontaneous emission is more predominant than stimulated emission, laser
action is NOT POSSIBLE at optical frequencies under thermal equilibrium.
12. For a semiconductor
laser, the bandgap is 0.9eV. What is the wavelength of light emitted from it.
Use the following data:
Solution
C
= 3 × 108 m/s.
λ
= 6.625 × 10‒34 Js
Given
Data
Eg = 0.9 eV = 0.9 × 1.6×10‒19
Joules
Formula:
Wavelength
λ = hc / Eg = (6.625×10‒34×3×108)
/ (0.9×1.6×10‒19)
λ=1.3802×10‒6m
The wavelength of light emitted = 1.3802 μm
13. For a
heterojunction semiconductor laser, the band gap of the semiconductor used is
1.44 eV. By doping, the band gap of the semiconductor is increased by 0.2 eV.
Calculate the change in the wavelength of the laser.
Solution
Given
Data
Eg1 = 1.44 eV = 1.44 × 1.6 × 10‒19
J
Eg1 = 2.309 × 10‒19 J
Eg2
= 1.44 + 0.2
Eg2
= 1.64 eV =
1.64 × 1.6 × 10‒19 J
Eg2
= 2.624 × 10‒19 J
Formula:
We
know
λ1
= hc / Eg1
λ2
= hc / Eg2
λ1
‒ λ2 = hc [ 1/Eg1
‒ 1/Eg2 ]

λ1
‒ λ2 = 6.625 × 10‒34 × 3 × 108 × ( 1/2.309×10‒19
‒ 1/2.624×10‒19 )
λ1
‒ λ2 = 1.9875 × 10‒25 × [4.33 × 1018 ‒ 3.81 ×
1018]
λ1
‒ λ2 = 1.987 × 10‒25 × [5.2 × 1017] m
λ1
‒ λ2 = 1033 × 10‒10 m
λ1
‒ λ2 = 1033Å
Change in Wavelength = 1033Å.
14. For a laser at 2.0
m distance from the laser output beam spot diameter is 6.0 mm and beam,
divergence is 1.2 mrad. Calculate the beam spot diameter at 5.0 m distance from
the laser output.
Solution
Given
Data
d2
= 5m
d1
= 2m
Diameter
D1 = 6 × 10−3 m
r1
= 3 × 10‒3 m
ϕ = 1.2 mrad.
Formula
We
know that ϕ = r2‒r1 / d2‒d1
ϕ
= r2‒r1 / d2‒d1
r2
= ϕ(d2‒d1) + r1
r2
= 1.2 × [5 – 2] + 3 × 10−3
r2
= 3.603 m.
Diameter
D2 = 2 × 3.603
D2
= 7.206m
Beam spot diameter at 5m = 7.206m.
ADDITIONAL SOLVED PROBLEMS
1. The first line of
the principal series of Sodium D‒line at 5890Å. This corresponds to a
transition from the first excited state to the ground state. What is the energy
in electron volts of the first excited state?
Solution
The
emitted energy E = hv = hc / λ
= (6.625×10‒34×3×108 ) /
(5890×10‒10)
= 3.3743 × 10‒19 Joules
= 3.3743×10‒19 / 1.602×10‒19
= 2.1063 eV.
The Energy of the first excited state = 2.1063 eV.
2. What is the ratio of
the stimulated emission to spontaneous emission at a temperature of 280°C for
Sodium D‒line?
Solution
Stimulated
emission / Spontaneous emission = R21(ST) / R21(SP)
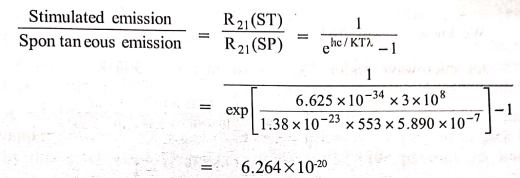
= 6.264 × 10‒20
The ratio between the stimulated emission and spontaneous
emission = 6.264 × 10‒20
3. A CO2
laser source emits light at a wavelength of 9.6 μm and has an output power of
10kW. How many photons are emitted in each hour by this laser while operating?
Solution
The
frequency of the photon emitted by the laser beam = c/ λ
V
= 3×108 / 9.6×10‒6
=
3.125 × 1013 Hz
We
know that, The energy of a photon E = hv
(or)
E = 6.625 × 10‒34 × 3.125 × 1013
(or)
E = 2.07 × 10‒20 J
Energy
emitted by the laser = 10 kilowatts.
=
10 × 103 W = 104 Js‒1
=
104 × 60 × 60 J hour‒1
The
number of photons emitted per hour = 104×60×60 / 2.07×10‒20
=
1.7391 × 1027 photons per hour
The CO2 laser source emits 1.739×1027
photons per hour.
4. Examine the
possibility of MASER action at thermal equilibrium using Einsteins theory of
light.
Solution
We
know
еhv/KBT
‒
1 = Rsp / Rst
for
microwave region λ = 10 cm ; T = 300° K
hv /
KBT = (6.625 × 10‒34
× 3 × 108) / (10×10‒2×1.38×10‒23×300)
Rsp
/ Rst = еhv/KBT
‒
1 = e(4.8 × 10‒4)
‒ 1
=
1.00048 ‒ 1
Rsp / Rst = 4.8 × 10‒4
Since
the spontaneous emission is lesser than stimulated emission, MASER action is
possible at thermal equilibrium.
5. An LED emits green
light of wavelength λ = 5511.11Å. Find out the value of Eg.
Solution
Eg
= hc / λ Joules
Eg
= [ hc / 1.602×10‒19λ
] eV
Eg
= [ 6.625×10‒34×3×108 / 1.602×10‒19λ ]
eV
Eg
= 12406.36×10‒10 / 5511.11×10‒10 eV
Eg = 2.25 eV
ASSIGNMENT PROBLEMS
1.
Calculate the ratio of the stimulated emission to spontaneous emission at a
temperature 300°C for the Sodium D‒line. (Ans:
2.919×10‒19)
2.
Calculate how many photons are emitted in each minute in a He‒Ne laser source,
which emits light at a wavelength of 6328Å. The output power of this source is
8mW. (Ans: 1.528×1018
photons/minute)
3.
A ruby laser source emits light at a wavelength of 6943Å and has an output
power of 105 watts. How many photons are emitted in each hour by
this laser while operating? (Ans:
1.257×1027 photons per hour)
4.
Transition occurs between a metastable state E3 and an energy state
E2, just above the ground state. If emission is at 1.1 μm and E2=0.4×10‒19
J, find the energy of the E3 state. (Ans: 2.2068×10‒19 J)
5.
If laser action occurs by the transition from an excited state to the ground
state E1 = 0, and if it produces light of 650 nm wavelength, what is
the energy level of the excited state. (Ans:
3.0576×10‒19 J)
6.
Given Eg=1.43 eV. Find the wavelength of the light emitted by the
diode for the given energy gap. Also find the colour of light emitted. (Ans: λ = 28671.32Å, IR)
Applied Physics I: Chapter 10: Applied Optics - Lasers : Tag: Applied Physics : - Lasers: Important Solved Problems
Applied Physics I: Chapter 10: Applied Optics - Lasers
Under Subject
Applied Physics I
PH25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Related Subjects
English Essentials I
EN25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
தமிழர் மரபு - Heritage of Tamils
UC25H01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Calculus
MA25C01 Maths 1 M1 - 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I
PH25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Applied Chemistry I
CY25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Makerspace
ME25C04 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming C
CS25C01 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Computer Programming Python
CS25C02 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Fundamentals of Electrical and Electronics Engineering
EE25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Mechanical Engineering
ME25C03 1st Semester | 2025 Regulation | 1st Semester 2025 Regulation
Introduction to Civil Engineering
CE25C01 1st Semester Civil Department | 2025 Regulation | 1st Semester 2025 Regulation
Essentials of Computing
CS25C03 1st Semester - AID CSE IT Department | 2025 Regulation | 1st Semester 2025 Regulation
Applied Physics I Laboratory
PH25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Applied Chemistry I Laboratory
CY25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming C Laboratory
CS25C01 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Computer Programming Python Laboratory
CS25C02 1st Semester practical Laboratory Manual | 2025 Regulation | 1st Semester Laboratory 2025 Regulation
Engineering Drawing
ME25C01 EEE Mech Dept | 2025 Regulation | 2nd Semester 2025 Regulation
Basic Electronics and Electrical Engineering
EE25C04 1st Semester ECE Dept | 2025 Regulation | 2nd Semester 2025 Regulation

















